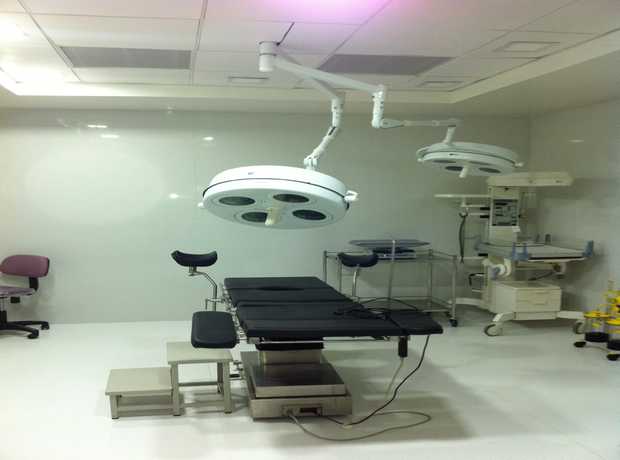
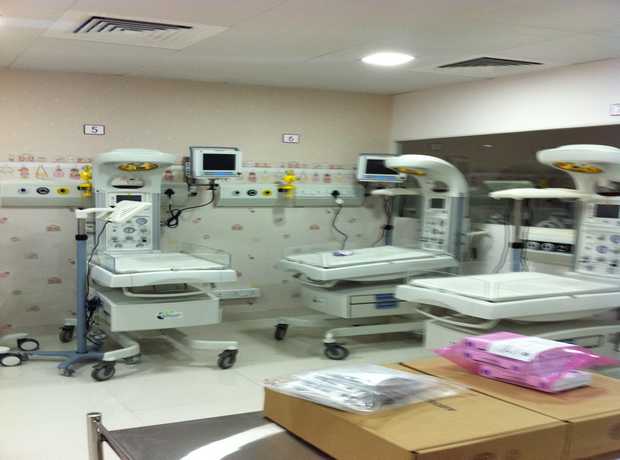
As Chicago continues its impressive urban expansion, particularly in the healthcare, pharmaceutical, and research sectors, the demand for advanced, compliant, and highly efficient sterilization and autoclave rooms is growing rapidly. These critical environments are the backbone of infection control and research integrity, making their construction a project of paramount importance. With ever-stricter regulations, evolving technological demands, and heightened user expectations, navigating the complexities of turnkey sterilization and autoclave room construction in Chicago requires specialized expertise that goes beyond general construction knowledge. This comprehensive guide is designed to walk you through the entire step-by-step process, from initial concept to final handover, ensuring your project not only meets but exceeds all necessary standards and delivers optimal performance for years to come.
The year 2025 marks a pivotal moment for construction in Chicago, with a focus on sustainable, smart, and highly specialized infrastructure. For facilities requiring rigorous sterilization processes, such as hospitals, diagnostic laboratories, pharmaceutical manufacturing plants, and advanced research institutions, the construction of a dedicated sterilization and autoclave room is a significant investment. This guide will delve deep into the nuances of planning, designing, constructing, and commissioning these vital spaces, highlighting why a turnkey approach is the most efficient and reliable path to success in the dynamic Chicago market.
Understanding Turnkey Sterilization and Autoclave Room Construction in Chicago
The concept of “turnkey construction” represents the ultimate end-to-end solution for any building project, but its value is amplified exponentially when applied to highly specialized facilities like sterilization and autoclave rooms. In a turnkey model, a single, experienced company takes full ownership and responsibility for every aspect of the project – from the earliest conceptual design and strategic planning through architectural blueprints, engineering, procurement, construction, installation, quality control, commissioning, and ultimately, the final handover. This integrated approach means you, the client, receive a complete, ready-to-operate facility with minimal stress and maximum efficiency.
For sterilization and autoclave rooms, where precision, stringent compliance with health regulations, and seamless integration of complex mechanical, electrical, and plumbing (MEP) systems are non-negotiable, a turnkey solution is not just a convenience but a strategic necessity. These rooms are intricate ecosystems designed to maintain sterility, manage biohazards, and operate sophisticated equipment. Any misstep in design or construction can have severe consequences, impacting safety, operational efficiency, and regulatory compliance. Therefore, entrusting the entire project to a single, accountable expert ensures that all elements are perfectly coordinated and executed.
Chicago, with its distinct building codes, diverse urban environment, specific environmental considerations, and a robust regulatory landscape, presents unique challenges and opportunities. A local expert with deep roots in Chicago construction is absolutely essential for navigating these intricacies. They possess not only the technical prowess but also the invaluable local knowledge of permitting processes, vendor networks, and specific compliance requirements that are unique to the city. This local expertise, combined with a turnkey methodology, provides an unparalleled advantage in delivering a successful, compliant, and high-performing sterilization and autoclave room.
Why Choose a Turnkey Solution for Your Chicago Project?
Opting for a turnkey construction solution for your critical sterilization and autoclave room in Chicago offers a multitude of benefits that traditional multi-vendor approaches simply cannot match. These advantages translate directly into reduced risk, optimized costs, and accelerated project timelines:
- Simplified Project Management: Imagine a complex orchestra with a single, expert conductor. That’s the essence of turnkey. You have one primary point of contact and one accountable entity for the entire project. This singular responsibility drastically streamlines communication channels, eliminates the common finger-pointing between different contractors, and significantly reduces the potential for costly errors, delays, and misunderstandings that often arise in projects with multiple stakeholders.
- Cost-Effective: A turnkey provider offers a comprehensive, often fixed-price proposal that encompasses all stages of the project. This predictability in costs, coupled with efficient workflows and integrated planning, minimizes the risk of budget overruns, change orders, and hidden expenses that can plague segmented projects. Integrated procurement and value engineering also contribute to significant cost savings without compromising quality.
- Compliance Assurance: Sterilization and autoclave rooms are subject to some of the strictest regulatory frameworks, including local Chicago building codes, fire safety regulations, health department mandates, and national healthcare standards (e.g., AAMI, CDC, OSHA). A specialized turnkey provider possesses expert, up-to-date knowledge of all these intricate requirements. They ensure every design choice and construction detail is compliant, providing you with the peace of mind that your facility will pass all inspections and operate safely and legally from day one.
- Faster Completion Times: The integrated nature of turnkey projects means that design, procurement, and construction phases can often overlap and run concurrently, rather than sequentially. This parallel processing, combined with efficient scheduling and a dedicated team, accelerates the overall construction timeline. For critical facilities, getting to operational readiness faster can mean earlier revenue generation or improved service delivery.
- Guaranteed Quality and Performance: With a single entity responsible for the entire project, there is a clear accountability for the final quality and performance of the facility. Turnkey providers stake their reputation on delivering a fully functional, high-performing asset that meets the client’s specifications and regulatory requirements. This often includes post-construction support and warranties.
Skydome Designs offers comprehensive turnkey solutions, managing everything from initial concept to final inspection for your critical Chicago facility. We ensure seamless integration, superior quality, and unwavering compliance. Learn more about our services and how we can bring your vision to life.
Step-by-Step Guide to Turnkey Sterilization and Autoclave Room Construction in Chicago
Constructing a state-of-the-art sterilization and autoclave room in Chicago is a meticulous process, demanding a structured, phased approach. Here’s a detailed breakdown of the key stages involved, highlighting the critical considerations at each juncture:
1. Project Planning and Design
This is arguably the most crucial initial phase, as it lays the entire foundation for the project’s success. It involves a deep dive into understanding your specific operational requirements, defining the project scope, and developing a highly detailed and compliant design plan.
- Needs Assessment and Feasibility Study: Before any lines are drawn, a thorough needs assessment is conducted. This involves understanding the specific function of the room (e.g., central sterile supply department for a hospital, research lab processing, pharmaceutical packaging sterilization). Key questions include: What types of materials will be sterilized? What is the expected volume and throughput? What specific equipment (e.g., steam autoclaves, EtO sterilizers, plasma sterilizers, washer-disinfectors) will be used, and what are their utility requirements (power, water, steam, drainage)? What are the current and future growth projections? This assessment also includes a site feasibility study to determine optimal location, spatial constraints, structural integrity, and existing utility infrastructure.
- Conceptual Design and Workflow Optimization: Based on the needs assessment, a preliminary conceptual design is created. This involves developing floor plans that optimize workflow – typically following a unidirectional “dirty to clean” flow to prevent cross-contamination. Consideration is given to receiving, decontamination, preparation and packing, sterilization, sterile storage, and distribution zones. Adjacencies to other departments (e.g., operating rooms, labs) are critically analyzed. Regulatory standards from organizations like AAMI (Association for the Advancement of Medical Instrumentation), CDC (Centers for Disease Control), OSHA (Occupational Safety and Health Administration), and local Chicago Department of Public Health are integrated from the very start to ensure the design inherently supports infection control and safety protocols.
- Detailed Design – Architectural, Structural, and MEP: This phase translates the conceptual design into comprehensive, buildable plans.
- Architectural Design: Specifies room layouts, material selections (e.g., non-porous, easily cleanable, chemical-resistant surfaces, seamless flooring), wall finishes, ceilings, doors (often interlocking for pressure differentials), pass-through windows, and specialized lighting (e.g., cleanroom-grade, anti-microbial). Attention is paid to creating a positive pressure environment in sterile areas and negative pressure in decontamination zones to control airborne contaminants.
- Structural Design: Ensures the building’s framework can support the substantial weight of autoclaves, water purification systems, and other heavy equipment. Vibration control is often a critical consideration, especially in multi-story buildings or research facilities, to maintain equipment precision. Seismic considerations are also factored into the structural design, given Chicago’s region.
- MEP (Mechanical, Electrical, Plumbing) Design: This is the lifeblood of the sterilization room. Detailed plans are developed for HVAC (heating, ventilation, and air conditioning) with HEPA filtration, precise temperature and humidity control, and air exchange rates; specialized plumbing for pure water systems (DI/RO water for autoclaves, softened water for washers), steam generation lines, and dedicated drainage for sterile processing and chemical waste; and robust electrical systems to power high-demand equipment, including dedicated circuits, emergency power backups (UPS/generators), and grounding.
- BIM (Building Information Modeling): Utilizing advanced BIM technology, Skydome Designs creates a 3D digital representation of the entire facility. This allows for accurate visualization, clash detection (identifying conflicts between architectural, structural, and MEP elements before construction begins), precise quantity take-offs, and enhanced coordination among all project stakeholders. BIM significantly reduces errors, minimizes rework, and improves project efficiency and predictability.
At Skydome Designs, our experienced team of architects, engineers, and healthcare planners works closely with you through every sub-phase to create a customized design that not only meets your exact operational needs but also exceeds regulatory compliance and future-proofs your investment. Contact us today for a consultation to discuss your project and discover how our meticulous planning and design approach can set your Chicago facility up for success.
2. Obtaining Permits and Approvals in Chicago
Navigating Chicago’s complex and multi-layered permitting process is a critical phase that demands local expertise and meticulous attention to detail. Delays in obtaining the necessary permits can significantly impact your construction timeline and budget. This step involves a series of submissions, reviews, and potential revisions with various city departments and regulatory bodies.
- Pre-Application Meetings: Often, an initial meeting with the Chicago Department of Buildings (DOB) and other relevant agencies (e.g., Department of Public Health) can clarify specific requirements, potential challenges, and streamline the application process. This proactive step helps identify unique project considerations early.
- Detailed Plan Submission: Comprehensive architectural, structural, MEP, and fire safety plans are meticulously prepared and submitted to the relevant authorities for review. These plans must demonstrate full compliance with the Chicago Building Code, zoning ordinances, fire codes, and specialized healthcare facility standards.
- Addressing Feedback and Revisions: It is common for city reviewers to request clarifications, revisions, or additional information. An experienced local partner can efficiently interpret this feedback, make the necessary adjustments to the plans, and resubmit them, ensuring a swift approval process. Understanding the nuances of what each department is looking for is paramount here.
- Securing All Necessary Building Permits and Approvals: This includes, but is not limited to:
- Building Permits: For the overall construction and structural modifications.
- Electrical Permits: For all electrical installations, including specialized power for autoclaves and safety systems.
- Plumbing Permits: For all water supply, drainage, and specialized pure water systems.
- HVAC Permits: For ventilation, air conditioning, and exhaust systems, particularly critical for sterile environments.
- Environmental Permits: Potentially required for waste disposal or specialized exhaust systems related to sterilization chemicals.
- Fire Safety Permits: Ensuring the design meets all fire suppression, alarm, and egress requirements for a healthcare or laboratory facility.
- Zoning and Land Use Approvals: Verification that the proposed construction aligns with Chicago’s zoning ordinances for the specific site. For new construction or significant renovations, this can sometimes involve additional public hearings or variances.
A delay in permits can cascade into significant construction delays and cost increases. Skydome Designs possesses extensive experience in navigating the intricacies of Chicago’s permitting landscape. Our in-house team is adept at preparing accurate submissions, proactively addressing potential issues, and efficiently communicating with city officials to expedite the approval process. We can help you navigate these complex processes, ensuring your project stays on track and compliant from the outset.
3. Construction and Installation
This phase transforms the meticulously designed plans into a tangible, functional sterilization and autoclave room. Adherence to the approved design plans, strict quality control, and unwavering compliance with building codes and safety regulations are paramount throughout this stage. This is where precision execution and robust project management come to the forefront.
- Site Preparation and Demolition (if applicable): The initial physical work begins with preparing the construction site. This can involve selective demolition of existing structures, site clearing, excavation for foundations or utility trenches, and ensuring the site meets all safety requirements, including temporary fencing, signage, and securing the perimeter. Comprehensive health and safety plans are implemented to protect workers and the surrounding environment, especially in active healthcare or research campuses.
- Structural Engineering and Framework: Implementing the structural framework of the room is a critical step. For sterilization rooms, this often involves specific considerations such as reinforced concrete slabs or steel structures designed to bear the significant weight of large autoclaves, water purification systems, and other heavy equipment. Vibration control measures may be integrated to ensure the stability and precision of sensitive instruments. Walls, floors, and ceilings are constructed to meet specific load-bearing requirements and provide a robust envelope for the sterile environment.
- MEP Systems Installation: The installation of Mechanical, Electrical, and Plumbing (MEP) systems is a highly specialized and intricate process for these facilities.
- Mechanical (HVAC): Installation of air handling units (AHUs), ductwork with integrated HEPA filtration, exhaust fans, and controls for maintaining precise temperature, humidity, and critical air pressure differentials (e.g., positive pressure in sterile storage, negative pressure in decontamination).
- Electrical: Laying out conduits, wiring, and installing panels for dedicated power circuits for autoclaves (often requiring high voltage and amperage), emergency power outlets, uninterrupted power supply (UPS) systems for critical controls, and specialized lighting systems.
- Plumbing: Installation of piping for softened water, deionized/reverse osmosis (DI/RO) water systems for autoclaves and washers, steam lines (if central steam is used), and dedicated drainage systems for both sanitary and potentially contaminated waste, ensuring no cross-contamination.
- Specialized Systems: This may also include medical gas lines (if applicable), data infrastructure for monitoring and control systems, and communication pathways.
Seamless integration and meticulous labeling of these complex systems are vital.
- Equipment Installation and Calibration: This phase involves the precise placement and secure installation of all specialized equipment:
- Autoclaves and Sterilizers: Steam sterilizers, ethylene oxide (EtO) sterilizers, hydrogen peroxide gas plasma sterilizers, and other advanced sterilization units are delivered, positioned, connected to utilities, and rigorously tested.
- Washer-Disinfectors: Automated washing equipment for instruments and glassware.
- Water Purification Systems: RO/DI units, water softeners, and associated storage tanks and distribution loops.
- Other Cleanroom Equipment: Laminar flow hoods, biological safety cabinets, specialized drying cabinets, and instrument reprocessing units.
Initial calibration and functional testing of each piece of equipment are performed in conjunction with the system installation.
- Finishing Touches and Cleanroom Envelope: The final interior finishes are applied, specifically chosen for their cleanroom and infection control properties. This includes:
- Flooring: Seamless, non-porous, chemical-resistant, anti-static, and easy-to-clean materials (e.g., epoxy, welded vinyl).
- Walls and Ceilings: Smooth, non-shedding, washable surfaces, often with integrated utilities and lighting. Modular cleanroom panels are frequently used for flexibility and superior finishes.
- Doors and Pass-Throughs: Installation of automatic, often interlocking, doors to maintain pressure differentials and prevent contamination. Pass-through windows and chambers are installed to transfer materials between different zones without compromising environmental control.
- Lighting: Installation of sterile-environment-grade, high-efficiency lighting with appropriate illumination levels.
- Environmental Monitoring Systems: Sensors for temperature, humidity, and pressure differentials are installed and integrated into the building management system.
Our extensive experience ensures that every aspect of the construction and installation phase is executed with the highest degree of professionalism and adherence to global best practices. We’ve delivered 463+ turnkey sterilization and autoclave room construction assignments across Chicago and globally over 12+ years. Our on‑time delivery rate is >98%, underpinned by multi‑disciplinary reviews and robust post‑occupancy support, ensuring exceptional outcomes for your critical facility.
4. Quality Control and Commissioning
The successful completion of a sterilization and autoclave room project hinges on rigorous quality control (QC) and a comprehensive commissioning process. This phase ensures that every component and system functions exactly as designed, meets all performance specifications, and complies with the strictest regulatory standards before the facility becomes operational.
- Thorough Testing of All Systems and Equipment: This involves a systematic verification of individual components and integrated systems:
- MEP System Testing: HVAC systems undergo air balance testing to verify airflow rates, pressure differentials, and filtration efficiency (e.g., HEPA filter integrity testing). Plumbing systems are pressure tested for leaks, and water quality is confirmed (e.g., DI/RO water purity). Electrical systems are tested for proper voltage, current, grounding, and emergency power transfer.
- Autoclave and Sterilizer Performance Testing: Each piece of sterilization equipment undergoes factory acceptance testing (FAT) and site acceptance testing (SAT). This includes leak tests, cycle efficacy tests (using biological and chemical indicators), temperature mapping, and pressure verification to ensure consistent and effective sterilization.
- Building Envelope Integrity: Air tightness tests may be performed to ensure the integrity of the cleanroom envelope, preventing uncontrolled air infiltration or exfiltration.
- Safety System Verification: Fire alarms, suppression systems, emergency lighting, and intercom systems are thoroughly tested for functionality and compliance.
- Verifying Compliance with All Relevant Standards and Regulations: During QC, every aspect is cross-referenced against design specifications, local Chicago building codes, fire codes, health department regulations, and specific industry standards (e.g., AAMI ST79 for steam sterilization, ISO 14644 for cleanrooms). This meticulous verification ensures the facility is fully compliant and safe for its intended use. Documentation of all tests and compliance checks is meticulously gathered.
- Commissioning the Room for Operational Use: Commissioning is the formal process of verifying that the entire facility, as an integrated system, performs according to the owner’s project requirements and design intent. This includes:
- Operational Qualification (OQ): Demonstrates that all equipment and systems operate correctly within their specified parameters under various operating conditions.
- Performance Qualification (PQ): Verifies that the entire integrated facility consistently performs its intended function effectively and reproducibly. For sterilization rooms, this means demonstrating consistent achievement of sterility levels.
- Validation Protocols: For highly regulated environments (e.g., pharmaceutical manufacturing), formal validation protocols (IQ/OQ/PQ) are strictly followed and documented to provide objective evidence that the process consistently produces a product or result meeting pre-determined specifications.
- Regulatory Inspections: Coordinating and facilitating final inspections by local authorities (e.g., Chicago Department of Public Health, Department of Buildings) to obtain the necessary occupancy permits and operational licenses.
At Skydome Designs, our commitment to quality extends through every phase, culminating in a rigorous QC and commissioning process. Our multi-disciplinary teams ensure that your sterilization and autoclave room in Chicago is not just built to spec, but validated for peak performance and absolute compliance.
5. Handover and Training
The final step in the turnkey construction process is not merely handing over the keys, but ensuring a smooth transition into full operational use. This involves providing you with a fully functional facility and equipping your staff with the knowledge and skills necessary to operate and maintain it safely and efficiently. This phase solidifies the long-term success and sustainability of your investment.
- Providing Detailed Documentation and Manuals: A comprehensive package of documentation is provided, serving as the facility’s operational bible. This includes:
- As-Built Drawings: Finalized drawings that reflect all changes made during construction, providing an accurate record of the installed systems.
- Operation and Maintenance (O&M) Manuals: Detailed guides for all installed equipment and systems, covering routine operation, preventative maintenance schedules, troubleshooting steps, and safety procedures.
- Warranties and Guarantees: Documentation for all equipment, materials, and workmanship, outlining terms and conditions.
- Calibration Records and Certificates: Proof that critical instruments and equipment have been accurately calibrated.
- Permits and Approvals: Copies of all final occupancy permits, licenses, and regulatory approvals.
- Training Materials: Customized guides and presentations used during staff training.
This documentation is essential for ongoing compliance, maintenance, and future modifications.
- Offering On-Site Training Sessions: Our expert team conducts thorough, hands-on training for your operational and maintenance staff. This covers:
- Equipment Operation: Detailed instruction on how to safely and effectively operate all autoclaves, sterilizers, washers, and water purification systems.
- System Monitoring: How to interpret readings from environmental monitoring systems (temperature, humidity, pressure) and building management systems.
- Routine Maintenance: Daily, weekly, and monthly maintenance tasks for equipment and facility systems, including filter changes, cleaning protocols, and minor adjustments.
- Safety Protocols: Comprehensive training on emergency procedures, chemical handling, infection control practices within the room, and lockout/tagout procedures for maintenance.
- Troubleshooting: Guidance on identifying and addressing common operational issues to minimize downtime.
Training can be tailored to different staff roles, ensuring everyone has the necessary knowledge.
- Providing Ongoing Support and Maintenance Services: The relationship doesn’t end at handover. A reputable turnkey provider offers continued support:
- Post-Occupancy Support: Addressing any teething issues, answering questions, and providing clarification during the initial operational period.
- Preventative Maintenance Contracts: Offering scheduled maintenance services to ensure the longevity, efficiency, and continuous compliance of the facility’s complex systems. This can include regular calibration, inspection, and servicing of equipment and HVAC systems.
- Emergency Support: Providing rapid response for unexpected breakdowns or critical issues.
- Upgrade and Expansion Planning: Acting as a trusted partner for future facility modifications, technology upgrades, or expansions.
Skydome Designs is committed to ensuring your team is fully prepared to maximize the investment in your new sterilization and autoclave room. Our comprehensive handover process and ongoing support guarantee long-term operational excellence and peace of mind.
Choosing the Right Chicago Construction Experts
The success of your specialized sterilization and autoclave room project in Chicago hinges significantly on selecting the right construction partner. This is not a task to be taken lightly, as the unique demands of these facilities require a very specific skillset and proven track record. To ensure your investment yields a high-performing, compliant, and durable asset, look for a company that demonstrates expertise across several critical areas:
- Proven Experience in Turnkey Sterilization and Autoclave Room Construction: General contractors might build great office buildings, but sterilization rooms are a different league. Prior experience specifically with sterile processing environments, cleanrooms, and laboratories is non-negotiable. Look for a portfolio that showcases similar projects and ask for client references. An experienced firm will understand the nuanced requirements of infection control, critical airflow, and specialized equipment integration.
- A Deep Understanding of Chicago Building Codes and Healthcare Regulations: Chicago’s regulatory landscape is intricate and constantly evolving. Your chosen partner must have an in-depth, up-to-date knowledge of local building codes, fire safety regulations, Department of Public Health standards, and federal mandates (like AAMI, CDC, OSHA) that specifically apply to healthcare and research facilities. Their ability to navigate the permitting and approval processes efficiently will be a major determinant of your project’s timeline and budget.
- A Team of Skilled Architects, Engineers, and Project Managers: A turnkey solution requires a multidisciplinary in-house team or a highly coordinated network of specialists. This includes architects experienced in sterile environment design, structural engineers familiar with heavy equipment loads and vibration control, MEP engineers with expertise in critical utility systems (pure water, steam, specialized HVAC), and project managers who can orchestrate complex schedules and budgets.
- A Commitment to Quality, Safety, and Customer Satisfaction: Quality should be evident in every aspect, from material selection to construction practices and finishing. Safety records and protocols are paramount, especially in construction near operational facilities. Furthermore, a client-centric approach, characterized by clear communication, transparency, and responsiveness, is vital for a smooth and successful partnership.
- Financial Stability and Bonding Capacity: Ensure your chosen partner has the financial stability to complete large-scale projects and sufficient bonding capacity to protect your investment.
- Certifications and Affiliations: Look for relevant certifications (e.g., LEED accreditation for sustainable building) and affiliations with industry organizations (e.g., ASHE, AAMI) which demonstrate a commitment to industry best practices and continuous improvement.
Skydome Designs boasts nearly 30 years of expertise across India and abroad, with an in-house team of architects, healthcare planners, and project managers dedicated to delivering exceptional results. Our award-winning designs prioritize client needs, functional excellence, and sustainability. We deliver projects on-time, on-budget, and to global standards, ensuring your Chicago facility is a benchmark for quality and efficiency. When you partner with us, you benefit from end‑to‑end delivery for turnkey sterilization and autoclave room construction — strategy, design, construction and handover in Chicago, supported by global design standards and local code expertise.
The Role of MEP Services in Autoclave Room Construction
MEP (Mechanical, Electrical, and Plumbing) services are not just components of a building; they are the highly specialized, interconnected lifeblood of any modern facility, and absolutely foundational for the functionality, safety, and compliance of sterilization and autoclave rooms. In these critical environments, the precise design, meticulous installation, and seamless integration of MEP systems are paramount. They control the air quality, manage water purity, deliver reliable power to sophisticated equipment, and ensure a safe operational environment. Any compromise in MEP design or execution can lead to contamination, equipment failure, safety hazards, and regulatory non-compliance.
Specific MEP Considerations for Sterilization Rooms
The unique demands of sterilization rooms necessitate a level of detail and specialization in MEP design that far exceeds standard commercial construction. Each element must be carefully considered to support the rigorous processes taking place.
- Ventilation (HVAC) Systems: This is perhaps the most critical mechanical aspect for maintaining a sterile environment and ensuring operator safety.
- Air Quality Control: HVAC systems must incorporate multi-stage filtration, including high-efficiency particulate air (HEPA) filters, to remove airborne contaminants, pathogens, and particulate matter.
- Pressure Differentials: Maintaining precise air pressure gradients is vital. Sterile storage areas typically require positive air pressure relative to adjacent corridors to prevent unfiltered air ingress, while decontamination areas often require negative pressure to contain hazardous aerosols and odors.
- Air Change Rates: High air change rates (ACH) are specified to rapidly dilute and remove airborne contaminants.
- Temperature and Humidity Control: Strict control over temperature and humidity is essential for equipment performance, material stability, and inhibiting microbial growth. High humidity can lead to condensation and microbial proliferation, while low humidity can generate static electricity.
- Exhaust Systems: Dedicated and often isolated exhaust systems are required for equipment like EtO sterilizers to safely vent hazardous gases to the outside, away from air intakes, in compliance with environmental regulations.
- Energy Efficiency and Redundancy: Designing for energy efficiency is important, but for critical facilities, redundancy (e.g., dual fans, backup chillers) is often incorporated to ensure continuous operation in case of equipment failure.
- Plumbing Systems: Water is a fundamental utility for sterilization, and its quality is paramount.
- Purified Water Systems: Autoclaves and washer-disinfectors require high-purity water (e.g., deionized (DI) or reverse osmosis (RO) water) to prevent mineral buildup on instruments and equipment, which can compromise sterilization efficacy and equipment longevity. This involves dedicated water purification units, storage tanks, and distribution loops.
- Softened Water: For initial washing phases and general utility, softened water may be required to prevent scale formation and improve detergent efficacy.
- Steam Generation: Reliable and consistent steam generation (either through central building steam or dedicated steam generators) is crucial for steam autoclaves. This involves robust piping, condensate return systems, and safety features.
- Drainage Systems: Separate drainage systems are often designed for sanitary waste and potentially contaminated or chemical waste from decontamination areas, with appropriate neutralization or containment measures where required, in compliance with Chicago’s waste management regulations.
- Cross-Contamination Prevention: Strict adherence to backflow prevention and cross-connection control measures is essential to protect the public water supply and prevent contamination within the facility.
- Electrical Systems: Sterilization equipment is power-hungry, requiring robust and reliable electrical infrastructure.
- Dedicated Circuits: Autoclaves, washers, and specialized water purification units require dedicated, often high-amperage, electrical circuits to ensure stable power delivery and prevent overloading.
- Emergency Power and UPS: Critical equipment and life safety systems must be connected to an emergency power supply (e.g., generator, UPS) to ensure operations continue uninterrupted during power outages, protecting patient safety and research integrity.
- Grounding and Bonding: Proper electrical grounding and bonding are essential for safety, preventing electrical hazards, and protecting sensitive electronic equipment from surges.
- Data Infrastructure: Integration of data cabling for monitoring sterilization cycles, environmental conditions, and connecting to building management systems (BMS).
- Specialized Lighting: Installation of cleanroom-grade, sealed light fixtures that are easy to clean and do not harbor contaminants.
- Building Automation Systems (BMS): Integrating all MEP systems into a central BMS allows for real-time monitoring and control of environmental parameters (temperature, humidity, pressure), equipment status, alarms, and energy consumption. This enhances operational efficiency, facilitates rapid response to deviations, and supports documentation for compliance.
At Skydome Designs, our in-house MEP engineers possess specialized knowledge in designing and implementing these complex systems for sterilization environments. We integrate these essential services seamlessly into your autoclave room design, ensuring optimal performance, safety, regulatory compliance, and energy efficiency. Our approach guarantees that your Chicago facility operates with unparalleled reliability and precision.
Key Challenges and Solutions in Chicago Turnkey Construction
Building in a major metropolitan area like Chicago presents a unique set of challenges, especially for specialized projects like turnkey sterilization and autoclave rooms. A local expert’s ability to anticipate and navigate these complexities is crucial for project success.
- Dense Urban Environment and Site Logistics:
- Challenge: Chicago’s urban core is densely packed, often leaving limited space for construction staging, material delivery, and equipment access. Noise ordinances and traffic congestion further complicate operations, especially when working near active hospitals or research labs.
- Solution: Skydome Designs employs meticulous logistical planning, including just-in-time material delivery, off-site prefabrication where possible, strategic scheduling for deliveries during off-peak hours, and close coordination with city traffic management. We implement robust site safety plans to minimize disruption to surrounding facilities and public access.
- Historic Preservation Requirements:
- Challenge: Many areas of Chicago feature historic buildings. If your project involves renovation or expansion of such a structure, it may trigger complex historic preservation reviews and require adherence to specific architectural and material guidelines, adding layers of complexity and potential cost.
- Solution: Our team has experience working with Chicago’s Department of Planning and Development (DPD) and historic preservation agencies. We conduct early assessments to identify potential historic impacts and develop design solutions that respect architectural heritage while integrating modern, compliant sterilization facilities.
- Navigating Multiple City Departments and Bureaucracy:
- Challenge: Obtaining permits and approvals in Chicago involves coordinating with numerous departments – Department of Buildings (DOB), Department of Public Health (DPH), Department of Transportation (CDOT), Fire Department, etc. Each has its own review process, requirements, and timelines.
- Solution: Our local expertise and established relationships with key city officials enable us to streamline the permitting process. We prepare comprehensive, accurate submissions, proactively address feedback, and manage inter-departmental coordination, minimizing delays and ensuring compliance at every stage.
- Labor Availability and Union Regulations:
- Challenge: Chicago has a strong union presence in the construction industry. Navigating union agreements, labor availability, and specific work rules can be complex and impact project scheduling and costs.
- Solution: Skydome Designs works with a network of experienced, union-affiliated subcontractors who understand the local labor landscape. Our project managers are skilled in labor relations and scheduling, ensuring efficient deployment of skilled trades while adhering to all collective bargaining agreements.
- Weather Considerations:
- Challenge: Chicago’s extreme weather patterns, from harsh winters with heavy snowfall and freezing temperatures to hot, humid summers, can significantly impact construction schedules, material handling, and worker safety.
- Solution: We incorporate weather contingency planning into our project schedules. This includes implementing cold-weather construction techniques, ensuring proper site protection and heating, and scheduling weather-sensitive tasks during optimal seasons. Our robust project management anticipates and mitigates weather-related risks.
- Stringent Healthcare and Laboratory Standards:
- Challenge: Beyond general building codes, sterilization rooms in Chicago must meet specialized standards for infection control, hazardous waste management, air quality, and safety protocols from bodies like AAMI, ASHRAE, CDC, and CAP.
- Solution: Our in-house team includes healthcare planners and engineers who are experts in these specific standards. We ensure that every design element, material selection, and installation practice is fully compliant, often exceeding minimum requirements to provide a truly state-of-the-art and safe facility.
By partnering with Skydome Designs, you gain a strategic ally deeply familiar with Chicago’s construction environment. Our proactive approach to these challenges ensures that your turnkey sterilization and autoclave room project is delivered smoothly, efficiently, and to the highest standards, regardless of the urban complexities.
Future Trends in Sterilization Room Design and Construction
The field of sterilization and sterile processing is constantly evolving, driven by advancements in technology, stricter infection control protocols, and a growing emphasis on sustainability and efficiency. Future-proofing your Chicago sterilization room means incorporating forward-thinking design and construction trends.
- Automation and Robotics Integration: The future of sterile processing involves increased automation, from robotic arms for instrument handling and sorting to automated guided vehicles (AGVs) for transport. Designing rooms with the infrastructure (e.g., floor load capacity, clear pathways, power for charging stations, data integration) to accommodate these technologies will enhance efficiency, reduce human error, and improve staff safety.
- Smart Building and IoT Integration: Sterilization rooms will become increasingly “smart,” utilizing IoT sensors to monitor critical parameters in real-time – temperature, humidity, air pressure, equipment status, and energy consumption. This data can be fed into a centralized Building Management System (BMS) for predictive maintenance, optimized energy use, and enhanced operational insights.
- Sustainable Design and Energy Efficiency: With a global push towards sustainability, future sterilization rooms will prioritize energy-efficient HVAC systems (e.g., heat recovery, variable air volume), LED lighting, advanced insulation, and water conservation measures (e.g., efficient water purification systems, greywater recycling where appropriate). LEED certification and other green building standards will become more prevalent, reducing operational costs and environmental impact.
- Modularity and Adaptability: The ability to adapt to future needs is crucial. Modular construction techniques, standardized layouts, and flexible utility connections can allow for easier expansion, reconfiguration, or technology upgrades without extensive downtime or costly renovations. This approach is particularly valuable in fast-paced research and healthcare environments.
- Advanced Infection Control Technologies: Beyond traditional filtration, future designs may incorporate emerging technologies like UV-C germicidal irradiation in HVAC systems or room disinfection cycles, self-cleaning surfaces, and advanced material science to further reduce microbial proliferation. Enhanced air curtain technology for doorways and advanced pass-through systems will further bolster containment.
- Enhanced Ergonomics and Human Factors: While automation increases, the role of human operators remains critical. Future designs will focus on improved ergonomics to reduce repetitive strain injuries, optimized lighting to minimize eye strain, and thoughtful spatial planning to enhance workflow efficiency and reduce fatigue, ultimately leading to a safer and more productive workforce.
- Data Analytics and Artificial Intelligence (AI): Leveraging AI to analyze sterilization cycle data, predict equipment failures, optimize maintenance schedules, and identify trends in contamination or efficiency will become standard practice, leading to continuous improvement in sterile processing operations.
Skydome Designs is at the forefront of these innovations, integrating future-ready solutions into our turnkey sterilization and autoclave room designs for Chicago projects. We help you build facilities that are not only compliant today but also prepared for the technological advancements and operational demands of tomorrow.
Benefits of Partnering with Skydome Designs for Your Chicago Project
When considering a critical investment like a turnkey sterilization and autoclave room, choosing the right partner is paramount. Skydome Designs stands apart through a unique blend of global expertise, local Chicago nuance, and a steadfast commitment to client success. Our nearly three decades of experience translate directly into tangible benefits for your project:
- Unrivaled Experience and Proven Track Record: With nearly 30 years of experience designing and constructing complex facilities across India and globally, we bring a wealth of knowledge to your Chicago project. Our portfolio includes 463+ turnkey sterilization and autoclave room construction assignments across Chicago and globally over 12+ years, a testament to our specialized capabilities.
- Exceptional On-Time and On-Budget Delivery: We understand that delays and cost overruns are unacceptable for critical facilities. Our rigorous project management methodologies ensure an on‑time delivery rate of >98%, coupled with meticulous budget control. This predictable execution protects your investment and allows for seamless integration into your operational plans.
- Integrated In-House Expertise: Our strength lies in our multidisciplinary in-house team. This includes seasoned architects specialized in healthcare and cleanroom design, expert healthcare planners who understand operational workflows and regulatory compliance, and dedicated project managers who orchestrate every detail. This integrated approach ensures seamless coordination, minimizes communication gaps, and fosters innovation from concept to completion.
- Award-Winning Designs Focused on Functionality and Aesthetics: While functionality and compliance are our top priorities, we believe that critical facilities can also be aesthetically pleasing and inspiring. Our award-winning designs consistently marry cutting-edge technology with thoughtful layouts, superior materials, and sustainable practices, creating environments that are both highly efficient and conducive to staff well-being.
- Client-Centric Approach and Post-Occupancy Support: Your needs are at the heart of everything we do. We engage in deep consultations to understand your specific requirements and build a partnership founded on transparency and responsiveness. Our commitment extends beyond handover, with robust post‑occupancy support that underpins long-term operational success and provides peace of mind.
- Global Design Standards with Local Code Expertise in Chicago: We bring a world-class perspective to every project, incorporating the latest international best practices in design, technology, and sustainability. Crucially, we pair this global vision with unparalleled local code expertise in Chicago, ensuring that every aspect of your facility is fully compliant with municipal regulations and local health standards.
- Multi-Disciplinary Reviews for Uncompromised Quality: Every project undergoes stringent multi‑disciplinary reviews at critical junctures. This rigorous internal process involves our architects, engineers, and healthcare planners scrutinizing every detail to identify potential issues, optimize performance, and ensure the highest standards of quality and safety are met.
Choose Skydome Designs for end‑to‑end delivery for turnkey sterilization and autoclave room construction — strategy, design, construction and handover in Chicago. We offer unparalleled global design standards with local code expertise in Chicago. Let us transform your vision into a high-performing, compliant, and future-ready facility.
FAQ: Turnkey Sterilization and Autoclave Room Construction in Chicago
Here are some frequently asked questions about turnkey sterilization and autoclave room construction projects in Chicago, offering further insights into common concerns:
What are the key considerations for designing a sterilization room in Chicago?
Designing a sterilization room in Chicago involves a comprehensive array of considerations. Paramount among these are adherence to stringent local building codes, fire safety regulations, and specific mandates from the Chicago Department of Public Health for infection control. Beyond regulatory compliance, key factors include optimizing workflow to maintain a “dirty to clean” flow and prevent cross-contamination, designing for precise environmental control (temperature, humidity, air pressure differentials) using advanced HVAC with HEPA filtration, ensuring robust utility infrastructure (pure water, steam, dedicated electrical circuits), selecting durable and easily cleanable materials, and planning for ergonomic and safe operation. Future expandability and integration of smart technologies are also increasingly vital considerations for long-term viability.
How long does it typically take to build a turnkey autoclave room in Chicago?
The timeline for building a turnkey autoclave room in Chicago can vary significantly, typically ranging from 6 months to 18 months. This duration is influenced by several factors: the size and complexity of the room, whether it’s a new build or a renovation within an existing structure, the scope of specialized equipment, the efficiency of the permitting process in Chicago, and any site-specific challenges (e.g., tight urban access, demolition requirements). For a typical mid-sized, dedicated facility, a well-managed project with a turnkey provider like Skydome Designs can often expedite the process. Proper, detailed planning and efficient project management are absolutely essential for timely execution, mitigating delays at every stage.
What are the typical costs involved in turnkey sterilization room construction?
The typical costs involved in turnkey sterilization room construction are highly variable, as they depend on the room’s size, the level of cleanroom classification, the complexity of MEP systems, the brand and capacity of sterilization equipment (autoclaves, washers, water purification systems), and the specific finishes required. Costs can range from several hundred thousand dollars for a smaller, specialized lab room to several million dollars for a large, integrated central sterile supply department in a hospital. A detailed, transparent cost estimate should always be provided during the initial planning and design phases. Skydome Designs prides itself on providing comprehensive and transparent cost estimates, ensuring no hidden fees and clear financial planning from the outset.
What permits are required for constructing an autoclave room in Chicago?
Constructing an autoclave room in Chicago necessitates securing a range of permits from various city departments. The primary permits include general building permits from the Chicago Department of Buildings (DOB), electrical permits for specialized power installations, plumbing permits for water purification systems, steam lines, and drainage, and HVAC permits for critical ventilation and air quality control systems. Additionally, specific fire safety permits are required, and depending on the scope, environmental permits might be needed for specialized waste handling or exhaust systems. If the facility is part of a healthcare institution, further approvals from the Chicago Department of Public Health are essential. Navigating these requires a deep understanding of local ordinances.
How can I ensure my sterilization room meets all regulatory requirements in Chicago?
Ensuring your sterilization room meets all regulatory requirements in Chicago demands a multi-pronged approach. Firstly, partner with experienced Chicago construction experts like Skydome Designs who possess an in-depth understanding of local building codes, fire regulations, and healthcare standards (e.g., AAMI, CDC, OSHA). Secondly, integrate regulatory compliance into every phase, from initial design (e.g., workflow, material selection, pressure differentials) through construction. Thirdly, implement rigorous quality control measures and thorough commissioning processes, including performance validation and continuous testing of all systems and equipment. Finally, ensure all required permits are obtained and that the facility passes all final inspections by relevant city and health authorities.
What are the benefits of a modular sterilization room construction in Chicago?
Modular sterilization room construction offers several compelling benefits, particularly in a dense urban environment like Chicago. These include significantly faster construction timelines due to parallel off-site fabrication and on-site assembly, superior quality control as modules are built in a controlled factory environment, and reduced site disruption and waste. Modular units can also offer greater design flexibility for future expansion or relocation, and potentially lower overall costs due to optimized manufacturing processes. This approach is ideal for projects with tight deadlines, limited site access, or those requiring rapid deployment.
How does Skydome Designs ensure infection control compliance in its sterilization room projects?
Skydome Designs prioritizes infection control compliance from the very first sketch. Our strategy includes: designing clear “dirty to clean” unidirectional workflows; implementing precise HVAC systems with HEPA filtration and pressure differentials to control airflow; specifying non-porous, seamless, and chemical-resistant materials for all surfaces (floors, walls, ceilings); designing for optimal equipment placement and maintenance access; integrating hands-free technologies and pass-through chambers; and ensuring all systems meet or exceed AAMI, CDC, OSHA, and local Chicago Department of Public Health standards. Our multi-disciplinary review process rigorously checks for compliance at every design and construction stage.
What kind of post-construction support is offered by Skydome Designs?
Skydome Designs believes in building lasting partnerships, and our commitment extends well beyond project handover. Our post-construction support includes comprehensive documentation (as-built drawings, O&M manuals, warranties), thorough on-site training for your operational and maintenance staff on all equipment and systems, and readily available assistance for any initial operational queries or troubleshooting. We also offer options for ongoing preventative maintenance contracts to ensure the long-term optimal performance and compliance of your facility, and are a trusted partner for future upgrades or expansions.
Are your staff certified in relevant cleanroom or healthcare construction standards?
Yes, our in-house team at Skydome Designs comprises highly qualified and certified professionals across various disciplines. Our architects and engineers hold relevant professional licenses, and our project managers possess extensive experience in healthcare and specialized facility construction. Many of our team members hold certifications in areas such as cleanroom technology, infection control risk assessment (ICRA), sustainable design (e.g., LEED accreditation), and project management methodologies, ensuring that your project benefits from industry-leading expertise and adherence to the highest professional standards.
Conclusion
Turnkey sterilization and autoclave room construction in Chicago is a complex, high-stakes endeavor that demands meticulous planning, specialized expertise, and an unwavering commitment to quality and compliance. These critical facilities are essential for safeguarding public health, advancing scientific research, and ensuring the integrity of medical procedures. By following this comprehensive step-by-step guide and partnering with experienced Chicago construction experts like Skydome Designs, you can confidently navigate every phase of your project, ensuring it is a resounding success.
Our approach ensures end‑to‑end delivery for turnkey sterilization and autoclave room construction — strategy, design, construction and handover in Chicago. We combine global design standards with unparalleled local code expertise, providing a seamless and efficient journey from concept to fully operational facility. Our extensive portfolio, with over 463+ turnkey sterilization and autoclave room construction assignments across Chicago and globally over 12+ years, speaks to our proven capability and reliability, underlined by an impressive on‑time delivery rate of >98%, robust multi‑disciplinary reviews, and dedicated post‑occupancy support that underpins exceptional outcomes.
Skydome Designs brings nearly 30 years of experience across India and abroad, with a dedicated in-house team of architects, healthcare planners, and project managers committed to delivering exceptional results. Our award-winning designs prioritize client needs, functional excellence, and sustainability, ensuring your project is delivered on-time, on-budget, and to global standards. Contact us today to discuss your upcoming project and discover how our specialized expertise can benefit you, transforming your vision into a state-of-the-art sterilization facility ready for 2025 and beyond.
Contact Us
Ready to get started on your state-of-the-art sterilization and autoclave room project in Chicago? Contact Skydome Designs today to leverage our nearly 30 years of global expertise and local Chicago knowledge. We are eager to discuss your specific requirements and demonstrate how our turnkey solutions can bring your vision to life, ensuring a compliant, efficient, and future-ready facility.
- Phone: +91 7299072144
- Email: info@skydomedesigns.com
- Website: Skydome Designs
Let Skydome Designs be your trusted partner for delivering excellence in specialized construction. We look forward to hearing from you.