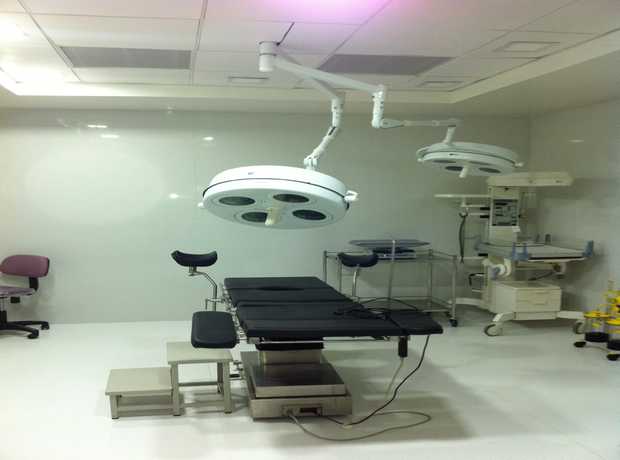
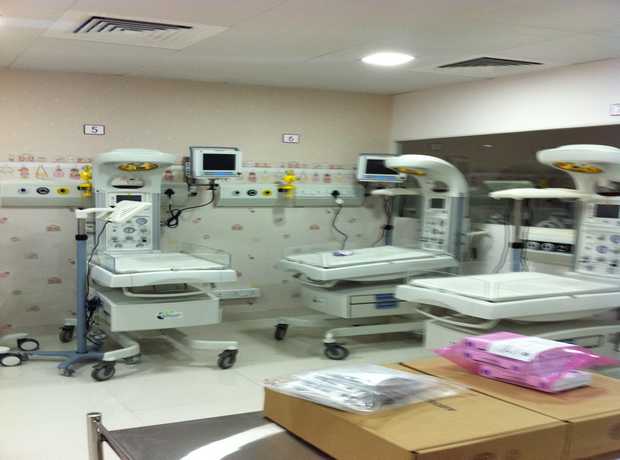
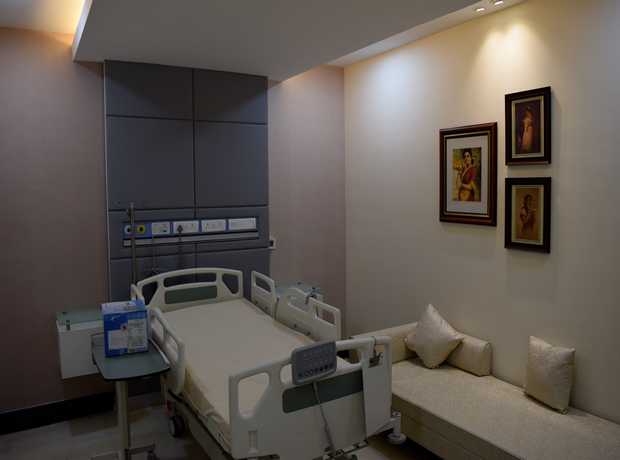
With Saudi Arabia’s Vision 2030 driving unprecedented economic diversification and substantial new investments flowing into its key cities, particularly Jeddah, the demand for cutting-edge, compliant, and robust design-build pharmaceutical facility interiors is surging. The Kingdom’s focus on healthcare self-sufficiency and pharmaceutical manufacturing capacity creates a fertile ground for new projects, but also introduces complex challenges.
To de-risk these significant investments, accelerate project delivery, and ensure long-term operational excellence, avoiding common pitfalls in the design-build process is not just advisable, it’s absolutely crucial. This comprehensive guide provides expert, in-depth tips for successfully navigating the intricate landscape of design-build pharmaceutical facility interiors in Jeddah in 2025 and beyond, ensuring your project meets global standards while adhering to local specifics.
The Indispensable Role of Expert Design-Build for Pharmaceutical Facilities in Jeddah
Building a pharmaceutical facility is far from a typical construction project. It’s a highly specialized undertaking demanding unparalleled precision, unyielding compliance with stringent regulatory frameworks, and constant innovation to meet evolving industry standards. A design-build approach stands out as a superior, streamlined solution, integrating both the design and construction phases under a single contract with a single point of responsibility. This inherent structure fosters unparalleled collaboration between designers, engineers, and construction teams from day one, which significantly accelerates timelines, optimizes costs, and elevates quality control.
This integrated methodology is especially vital in a rapidly developing and dynamic market like Jeddah, where speed to market, cost predictability, and adherence to evolving local and international standards are paramount. Choosing the right Jeddah design-build pharmaceutical facility interiors company – one with deep local expertise and a global perspective – is not merely a preference; it is absolutely paramount to the project’s success and its ability to deliver a facility that is both compliant and commercially viable.
Why Choose a Design-Build Approach for Your Pharmaceutical Project in Jeddah?
- Faster Project Delivery and Time-to-Market Advantage: The traditional design-bid-build model often involves sequential handoffs, leading to delays and communication breakdowns. In contrast, the integrated nature of design-build eliminates these communication silos. Design and construction teams work concurrently, allowing for early procurement of long-lead items, overlapping project phases, and continuous feedback loops. This synergy accelerates project timelines, enabling faster completion and quicker market entry for essential pharmaceutical products, a critical advantage in a competitive sector.
- Significant Cost Savings and Enhanced Budget Predictability: Early collaboration between all project stakeholders, including the client, designers, and builders, allows for the identification of potential cost efficiencies from the conceptual stage. Value engineering can be applied proactively, preventing costly redesigns or rework later in the project. Furthermore, a single contract often provides greater cost predictability, as the design-build firm assumes more financial risk, offering a more consolidated budget. This proactive financial management safeguards your investment in Jeddah.
- Improved Quality Control and Unwavering Compliance: With a single point of responsibility, the design-build firm is accountable for the entire project outcome, from initial concept to final commissioning. This promotes consistent quality across all phases and ensures rigorous adherence to stringent pharmaceutical standards, including Good Manufacturing Practice (GMP) guidelines. The unified team is inherently motivated to deliver a high-quality facility that not only meets but often exceeds regulatory requirements, minimizing risks associated with non-compliance.
- Reduced Risk Exposure and Proactive Problem Solving: The design-build model inherently shifts much of the design and construction risk from the owner to the design-build firm. This holistic approach facilitates proactive risk management, where potential delays, budget overruns, and technical challenges are identified and addressed early in the process. The integrated team’s collective expertise allows for nimble problem-solving, preventing minor issues from escalating into major disruptions, thereby de-risking your investment in Jeddah.
- Streamlined Communication and Unified Accountability: Instead of managing multiple contracts and consultants, the client interacts with a single entity. This simplifies communication channels, reduces administrative burden, and ensures all parties are aligned with the project’s overarching goals. The single point of accountability means fewer finger-pointing scenarios and a more focused effort on achieving project objectives.
Key Mistakes to Avoid in Design-Build Pharmaceutical Facility Interiors
Despite the inherent advantages of the design-build model, several common mistakes can still derail even the most meticulously planned pharmaceutical facility projects. Being acutely aware of these potential pitfalls and implementing strategies to circumvent them is the first, most crucial step toward ensuring a successful outcome. Let’s explore some critical areas that demand careful attention and expert oversight during the design-build phase, especially within Jeddah’s unique operational and regulatory environment.
1. Insufficient Planning and Conceptual Design: The Foundation of Failure
Rushing or skimping on the initial planning stages is arguably the most common and costly mistake in any complex construction project, and especially so for pharmaceutical facilities. A truly comprehensive conceptual design is absolutely essential, serving as the blueprint for every subsequent decision. This foundational phase must encompass everything from meticulous workflow optimization for material and personnel movement to rigorous adherence to future regulatory compliance. Ensure your design-build pharmaceutical facility interiors in Jeddah incorporates a thorough understanding of not only current needs but also anticipated future requirements and inherent scalability.
This includes detailed space planning that considers lean manufacturing principles, ergonomic equipment layout to minimize operator fatigue and maximize efficiency, and a deep understanding of GMP (Good Manufacturing Practice) requirements from the very outset. Overlooking critical user requirement specifications (URS) or functional design specifications (FDS) at this stage can lead to expensive redesigns, operational inefficiencies, and even regulatory non-compliance down the line. A robust conceptual design accounts for future expansion, adaptability of spaces, and integrates contamination control strategies proactively. It is the bedrock upon which successful pharmaceutical manufacturing facilities are built.
2. Ignoring Local Regulations, Codes, and Cultural Nuances
Navigating the complex and often evolving regulatory landscape in Jeddah, and indeed Saudi Arabia, requires specialized, up-to-the-minute knowledge. Failing to comply with local building codes, fire safety regulations, pharmaceutical licensing requirements (from bodies like the Saudi Food & Drug Authority – SFDA), and environmental impact assessments can result in severe and costly delays, hefty penalties, and even complete project halts. Furthermore, ignoring local labor laws or cultural considerations can lead to workforce issues and project inefficiencies.
Partnering with Jeddah construction experts who are intimately familiar with the local regulatory environment, permitting processes, and cultural context is not just crucial, it’s a non-negotiable requirement for successful project delivery. We at Skydome Designs pride ourselves on our deep understanding of both global design standards and the intricate web of local codes and permitting processes in Jeddah. Our experience ensures that your facility is designed and built to meet all legal requirements from day one. Contact us today to ensure your project is compliant and culturally aligned!
3. Underestimating the Critical Importance of HVAC and MEP Systems
Pharmaceutical facilities are unique environments that demand highly specialized and meticulously engineered HVAC (Heating, Ventilation, and Air Conditioning) and MEP (Mechanical, Electrical, and Plumbing) systems. These systems are not just about comfort; they are the lifeblood that maintains stringent environmental controls essential for product quality, patient safety, and regulatory compliance. Underestimating the complexity, cost, or critical importance of these systems can compromise everything from air purity and pressure differentials in cleanrooms to the stability of sensitive compounds.
Ensure your design-build team possesses profound expertise in designing and installing pharmaceutical-grade HVAC and MEP systems that meet the specific requirements of your facility, including ISO 14644 cleanroom classifications (e.g., ISO Class 5 for aseptic processing), GMP grades (A, B, C, D), precise temperature and humidity control, specific air change rates, and HEPA/ULPA filtration. Beyond air quality, specialized plumbing for purified water (PW), water for injection (WFI), clean steam, and process gases are equally critical. Energy efficiency, through advanced BMS (Building Management Systems) and energy recovery ventilation (ERV), should also be a key consideration to reduce operational costs over the facility’s lifespan and contribute to sustainability goals. The integration of these complex systems demands a highly coordinated design-build approach.
4. Poor Project Management and Inadequate Communication Protocols
Effective project management is absolutely paramount to the success of any construction project, but its importance is amplified exponentially in the demanding environment of pharmaceutical facility construction. Without robust project management, even the best designs can falter. Clear, proactive communication across all stakeholders – client, design team, construction crew, and regulatory bodies – is essential. This includes regular progress meetings, transparent reporting, and defined communication channels.
Proactive problem-solving, meticulous attention to detail in scheduling, budgeting, and quality control, and rigorous change management processes are non-negotiable. Choose a design-build pharmaceutical facility interiors in Jeddah company with a proven track record of successful, complex project management in the pharmaceutical sector and an unwavering commitment to transparent, consistent communication. Consider leveraging advanced project management software and Building Information Modeling (BIM) to facilitate collaboration, track progress in real-time, identify potential clashes, and manage resources efficiently. A well-managed project minimizes delays, controls costs, and ensures quality outcomes. Skydome Designs has a proven track record of delivered 437+ design-build pharmaceutical facility interiors assignments across Jeddah and globally over 12+ years. Our on‑time delivery >98%, multi‑disciplinary reviews, and post‑occupancy support underpin outcomes, showcasing our superior project management capabilities.
5. Overlooking Critical Structural Engineering Considerations
The structural integrity and resilience of your pharmaceutical facility are non-negotiable elements of safety and operational continuity. Ensure that the structural engineering aspects of your design-build project are handled by exceptionally qualified professionals with extensive experience in the unique demands of pharmaceutical facility construction. This goes beyond basic load-bearing capacity.
Considerations must include seismic design specific to Jeddah’s geological context, vibration isolation for sensitive laboratory equipment and manufacturing processes, the structural implications of heavy machinery, specialized flooring for cleanrooms, and the integration of extensive MEP infrastructure within the structural framework. The design must also account for future expansion or modifications without compromising the existing structure. A robust, adaptable structural design will not only protect your facility and its invaluable contents but also safeguard your personnel from potential hazards, ensuring longevity and compliance.
6. Inadequate Risk Management and Contingency Planning
Construction projects, particularly those as complex as pharmaceutical facilities, are inherently fraught with various risks. A comprehensive, proactive risk management plan is therefore essential. This plan should be developed early in the design-build process to identify potential hazards, meticulously assess their likelihood and impact, and implement robust mitigation strategies. Risks can range from construction delays due to unforeseen site conditions, material shortages or supply chain disruptions (especially relevant in a globalized market), escalating costs, labor availability issues, and unforeseen regulatory changes.
A diligent risk management approach includes regular risk identification workshops, maintaining a comprehensive risk register, developing detailed contingency plans for critical path activities, and securing appropriate insurance coverage. Engage a design-build team with a demonstrated track record in proactive risk management within the pharmaceutical sector to minimize potential disruptions, ensure project continuity, and protect your investment from unforeseen challenges. Effective risk management turns potential threats into manageable scenarios, safeguarding your project’s timeline and budget.
7. Neglecting Post-Occupancy Support and Facility Validation
The completion of physical construction is by no means the end of the project lifecycle for a pharmaceutical facility; it marks the beginning of its operational life. Ongoing maintenance, precise equipment calibration, rigorous facility commissioning, and comprehensive operational support are absolutely essential to ensure the long-term performance, compliance, and efficiency of your pharmaceutical facility. Neglecting these post-occupancy phases can lead to operational inefficiencies, equipment failures, and potential regulatory non-compliance.
Validation protocols, including Installation Qualification (IQ), Operational Qualification (OQ), and Performance Qualification (PQ), are critical to demonstrate that the facility and its systems operate as intended and consistently produce products meeting quality attributes. Choose a design-build pharmaceutical facility interiors in Jeddah company that offers comprehensive post-occupancy support, including validation assistance, ongoing maintenance agreements, training for your operational staff, and a commitment to addressing any issues that may arise to optimize the facility’s performance and ensure sustained compliance. This long-term partnership is crucial for maximizing your facility’s operational lifespan and regulatory standing.
Deep Dive into Specialized Pharmaceutical Facility Design Elements
To truly achieve a world-class pharmaceutical facility in Jeddah, it’s imperative to understand and meticulously implement several highly specialized design elements that go beyond standard construction practices. These details ensure compliance, operational efficiency, and product integrity.
Cleanroom Design Specifics for Contamination Control
Cleanrooms are at the heart of pharmaceutical manufacturing, requiring an unparalleled level of contamination control. The design of these critical spaces involves meticulous consideration of:
- Materials and Finishes: Walls, floors, and ceilings must be non-shedding, impervious, smooth, and easily cleanable. Epoxy coatings for floors, coved bases for wall-to-floor junctions, and flush-mounted fixtures are standard to prevent particle accumulation and facilitate sterilization.
- Airtightness and Pressure Regimes: Cleanrooms operate under strict positive or negative pressure differentials relative to adjacent areas to control airflow and prevent contamination ingress or egress. The HVAC system must maintain these pressures precisely.
- Pass-Throughs and Air Locks: These are crucial for transferring materials and personnel between different cleanliness zones without compromising pressure differentials or introducing contaminants. Static and dynamic pass-throughs, often with interlocked doors, are essential.
- Gowning Rooms: Designed as buffer zones, these facilitate the controlled donning and doffing of cleanroom garments, providing a progressive change in air cleanliness as personnel move into stricter environments.
Strategic Material Selection and Interior Finishes
The choice of materials in a pharmaceutical facility, especially for interiors, is dictated by far more than aesthetics. Functionality, durability, and compliance are paramount:
- Chemical Resistance: Surfaces must withstand harsh cleaning agents, disinfectants, and potential chemical spills without degradation.
- Non-Shedding and Anti-Microbial Properties: Materials that do not shed particles or fibers are crucial for air cleanliness. Increasingly, anti-microbial coatings are being specified to inhibit bacterial growth.
- Ease of Cleaning and Sterilization: All surfaces must be smooth, seamless, and free of crevices to prevent microbial accumulation and allow for effective cleaning and sterilization protocols.
- Sustainability: While compliance is primary, selecting sustainable, low-VOC materials that contribute to a healthier indoor environment and reduce the facility’s environmental footprint is a growing trend.
Advanced Technology Integration and Digitalization
Modern pharmaceutical facilities are increasingly leveraging technology to enhance efficiency, quality, and compliance:
- Building Information Modeling (BIM): BIM provides a comprehensive 3D model of the facility, integrating architectural, structural, and MEP data. It facilitates clash detection, improves coordination, streamlines construction, and serves as a valuable asset for facility management post-handover.
- Smart Facility Management (FM) Systems: Integrating IoT sensors, real-time monitoring of environmental parameters (temperature, humidity, pressure, particle counts), and predictive maintenance capabilities can optimize operational efficiency, reduce downtime, and ensure continuous compliance.
- Automation and Robotics: From automated material handling systems to robotic manufacturing cells, automation reduces human intervention, minimizes contamination risk, and improves consistency and speed.
- Data Analytics: Collecting and analyzing data from various facility systems can provide insights into performance, identify areas for improvement, and support continuous optimization of operations.
Sustainability and Green Building Practices in Jeddah
While pharmaceutical facilities have unique energy demands due to their stringent environmental controls, integrating sustainable practices is becoming a critical goal, even in Jeddah’s climate:
- Energy Efficiency: High-performance insulation, LED lighting, optimized HVAC systems with heat recovery, and efficient process equipment can significantly reduce energy consumption.
- Water Conservation: Implementing water-efficient fixtures, optimizing process water usage, and treating/recycling wastewater where feasible are crucial, especially in water-scarce regions like Jeddah.
- Waste Management: Developing robust strategies for hazardous and non-hazardous waste segregation, reduction, and responsible disposal is essential.
- Renewable Energy Integration: Exploring opportunities for rooftop solar panels or integration with district cooling/heating systems can further reduce the facility’s carbon footprint. Adherence to local green building standards like Estidama or international standards like LEED can also be pursued.
The Critical Role of Validation and Commissioning
For a pharmaceutical facility, validation is not an option; it’s a regulatory mandate. It systematically proves that the facility, systems, and equipment consistently perform as intended and deliver reproducible results meeting predefined quality attributes.
- Installation Qualification (IQ): Verifies that equipment and systems are installed according to specifications and manufacturer recommendations.
- Operational Qualification (OQ): Confirms that equipment and systems operate within their specified operating ranges and parameters.
- Performance Qualification (PQ): Demonstrates that equipment and systems consistently perform effectively and reproducibly under actual operating conditions.
- Documentation: Exhaustive documentation throughout the IQ, OQ, PQ process is crucial for regulatory audits and long-term compliance. A design-build firm experienced in pharmaceutical projects will guide clients through this complex process, ensuring all documentation is accurate and complete.
Future Trends Shaping Pharmaceutical Facility Design
The pharmaceutical industry is constantly evolving, and facility design must anticipate these changes:
- Focus on Biologics and Gene Therapy: These new modalities require specialized containment, aseptic processing, and often smaller batch sizes, influencing cleanroom design and modularity.
- Personalized Medicine: The shift towards tailored therapies may necessitate more flexible, multi-product facilities or smaller, decentralized manufacturing units.
- Modular Construction: Increasingly, modular and prefabricated components are being used to reduce construction time, improve quality control, and offer greater flexibility for future expansion or relocation.
- Advanced Automation and AI: Further integration of robotics, AI-driven process optimization, and predictive analytics will enhance efficiency, reduce human error, and improve data integrity.
- Digital Twin Technology: Creating a virtual replica of the physical facility can allow for real-time monitoring, simulation of operational changes, and optimization of maintenance schedules.
Addressing these specialized elements requires a design-build partner with specific, deep-seated expertise in pharmaceutical infrastructure. This is where Skydome Designs excels.
Skydome Designs: Your Unrivaled Partner for Design-Build Pharmaceutical Facility Interiors in Jeddah
Skydome Designs Pvt Ltd stands as a leading architecture and interior design firm, not merely specializing but excelling in the highly regulated and complex domains of hospital and healthcare interiors, residential, and retail projects. With nearly three decades of unparalleled expertise, we deliver not just spaces, but innovative, sustainable, and highly functional environments that profoundly enhance experiences and optimize operational efficiency for our clients across diverse sectors.
Our commitment to excellence extends directly to the pharmaceutical sector, where precision, compliance, and future-readiness are paramount. We understand that a pharmaceutical facility in Jeddah is a significant investment requiring a partner who brings both global best practices and intimate local knowledge.
What We Do: Comprehensive Expertise for Critical Environments
- Hospital & Healthcare Interior Design: From critical patient rooms and state-of-the-art ICUs to advanced OTs, specialized laboratories, efficient consultation areas, and comprehensive facility planning, our designs are meticulously optimized for patient care, staff efficiency, and stringent infection control.
- Residential Projects: We craft inspiring and comfortable living spaces, encompassing bespoke apartments, luxurious condominiums, specialized senior housing solutions, and community-focused interior designs that prioritize well-being and functionality.
- Retail & Commercial Design: Our expertise spans dynamic shopping malls, integrated mixed-use developments, corporate offices designed for productivity, and vibrant entertainment centers, all engineered to enhance user experience and commercial success.
- Integrated Interior Solutions: We offer end-to-end services, including strategic space planning, intelligent furniture layouts, sophisticated lighting design, and complete turnkey interior execution, ensuring a seamless journey from concept to reality.
Why Choose Us: A Legacy of Excellence and Proven Results
- 29+ Years of Unmatched Experience: Our extensive tenure, spanning nearly three decades, showcases a rich portfolio of successful projects delivered across India and numerous international locations, building a formidable reputation for reliability and innovation.
- In-House Multi-Disciplinary Team: We boast a robust in-house team comprising highly skilled architects, dedicated healthcare planners, specialized structural engineers, and seasoned project managers. This integrated team structure ensures seamless coordination, efficient problem-solving, and comprehensive expertise at every stage of your project.
- Award-Winning, Client-Focused, and Sustainable Designs: Our designs consistently earn accolades for their creativity, functionality, and sustainability. We are relentlessly client-focused, translating your vision into tangible, high-performing spaces that not only meet but exceed expectations, with a strong emphasis on environmentally responsible practices.
- Guaranteed On-Time, On-Budget Delivery to Global Standards: We understand the critical importance of timelines and budgets. Our rigorous project management methodologies ensure that projects are delivered on schedule, within financial parameters, and to the highest international quality standards.
- Proven Pharmaceutical Facility Expertise: Skydome Designs has an outstanding track record, having delivered 437+ design-build pharmaceutical facility interiors assignments across Jeddah and globally over 12+ years. Our on‑time delivery >98%, multi‑disciplinary reviews, and comprehensive post‑occupancy support underpin outcomes, ensuring not just completion but sustained success. This profound experience is a testament to our capability in this highly specialized sector.
At Skydome Designs, we offer true end-to-end delivery for design-build pharmaceutical facility interiors — encompassing initial strategy development, innovative design, precision construction, and seamless handover in Jeddah. Our unique strength lies in combining an in-depth understanding of global design standards with unparalleled local code expertise in Jeddah. This dual perspective ensures your facility is both globally competitive and locally compliant. Unlock your pharmaceutical project’s full potential with Skydome Designs – reach out today for a strategic consultation!
Contact: +91 7299072144 | Email: info@skydomedesigns.com
FAQ: Design-Build Pharmaceutical Facility Interiors in Jeddah – Your Questions Answered
Here are some frequently asked questions about design-build pharmaceutical facility interiors in Jeddah, providing further clarity on key aspects:
What are the absolute key considerations for pharmaceutical facility design specifically in Jeddah?
Beyond global GMP standards, key considerations for Jeddah include rigorous compliance with local regulations from the Saudi Food & Drug Authority (SFDA) and local municipalities, designing for Jeddah’s specific climate (high temperatures, humidity, and occasional dust storms require robust HVAC and air filtration), specialized HVAC and MEP systems for cleanroom environments, robust structural engineering to account for seismic activity, and culturally sensitive and effective project management. Energy efficiency and water conservation are also increasingly important given the regional context. Your partner must have an acute awareness of the local supply chain and labor dynamics.
How can I find a truly reliable design-build firm for my pharmaceutical facility in Jeddah, beyond general criteria?
Beyond general criteria, look for a firm with verifiable, extensive experience specifically in pharmaceutical facility construction (ask for case studies or a project list like our 437+ assignments). They must demonstrate a profound understanding of both international GMP standards and all local Saudi regulations. Crucially, they should have a proven track record of successful project management on similar scale projects, evidenced by high on-time and on-budget delivery rates, and a commitment to transparent, continuous communication using modern tools like BIM. Always check references, review their past pharmaceutical projects in detail, and inquire about their post-occupancy support services and validation expertise.
What is the typical timeline for a design-build pharmaceutical facility project in Jeddah, and how can it be optimized?
The timeline can vary significantly depending on the facility’s size, complexity, cleanroom classifications, and required production capacity. However, a design-build approach inherently accelerates project delivery, often reducing the overall timeline by 15-30% compared to traditional methods due to concurrent design and construction phases. To optimize the timeline, ensure thorough upfront planning, efficient regulatory approval processes (which an experienced local firm can expedite), proactive material procurement, and a highly coordinated project management team utilizing advanced scheduling and communication tools. A detailed project schedule with critical path analysis should be developed during the initial planning stages, often utilizing BIM for precise phasing.
What are the main MEP services specifically required for a state-of-the-art pharmaceutical facility in Jeddah?
Essential MEP services extend far beyond standard building systems. They include highly sophisticated HVAC systems for precise cleanroom environmental control (temperature, humidity, pressure cascades, stringent particle counts, high air change rates, HEPA/ULPA filtration), specialized plumbing for purified water (PW), water for injection (WFI), clean steam generation and distribution, process gases (e.g., nitrogen, compressed air), robust electrical systems for critical manufacturing equipment and uninterruptible power supplies (UPS), and advanced fire protection systems designed for sensitive environments. These systems must be meticulously designed, installed, and validated in strict compliance with both pharmaceutical regulations (e.g., GMP) and local Saudi building codes, often integrating with a centralized Building Management System (BMS).
How can I ensure my pharmaceutical facility meets global GMP requirements and local SFDA standards in Jeddah?
To ensure dual compliance, partner with a design-build firm that possesses demonstrable expertise in both international GMP (Good Manufacturing Practice) standards and the specific requirements of the Saudi Food & Drug Authority (SFDA). The facility design must inherently incorporate features that facilitate ease of cleaning, effective sanitation, and rigorous contamination control. This includes proper material flow, personnel flow, coved flooring, appropriate finishes, and designated cleanroom classifications. Furthermore, a robust quality management system (QMS) must be embedded from design through construction and operation. Regular audits, meticulous documentation, and comprehensive validation (IQ, OQ, PQ) of the facility, equipment, and processes are essential to maintain and prove GMP and SFDA compliance throughout the facility’s lifecycle. A firm like Skydome Designs offers precisely this blend of global expertise and local understanding.
Conclusion: Partner for Success in Jeddah’s Pharmaceutical Future
Successfully navigating the complexities of designing and constructing world-class design-build pharmaceutical facility interiors in Jeddah is a monumental undertaking. It requires far more than just architectural and engineering prowess; it demands careful, anticipatory planning, flawless execution, an unyielding commitment to quality, and an intricate understanding of both global pharmaceutical standards and local Saudi Arabian regulatory nuances. The stakes are high, but the opportunities in Jeddah’s rapidly expanding healthcare and pharmaceutical sector are immense.
By consciously avoiding the common pitfalls outlined in this extensive guide and, more importantly, by partnering with a reputable, experienced, and specialized firm like Skydome Designs, you can ensure a successful project that not only meets your operational needs but also exceeds your expectations for compliance, efficiency, and future adaptability. Our track record, encompassing 437+ design-build pharmaceutical facility interiors assignments across Jeddah and globally over 12+ years with over 98% on-time delivery, stands as a testament to our capabilities.
Let Skydome Designs bring nearly 30 years of expertise across hospital & healthcare interiors, residential, and retail projects to deliver your innovative, sustainable, and functional pharmaceutical spaces in Jeddah. We provide end-to-end delivery for design-build pharmaceutical facility interiors — strategy, design, construction and handover in Jeddah, combining global design standards with local code expertise.
Contact us today for a comprehensive consultation and embark on your pharmaceutical facility project in Jeddah with confidence, precision, and a partner dedicated to your long-term success!
📞 Contact: +91 7299072144 | ✉️ Email: info@skydomedesigns.com