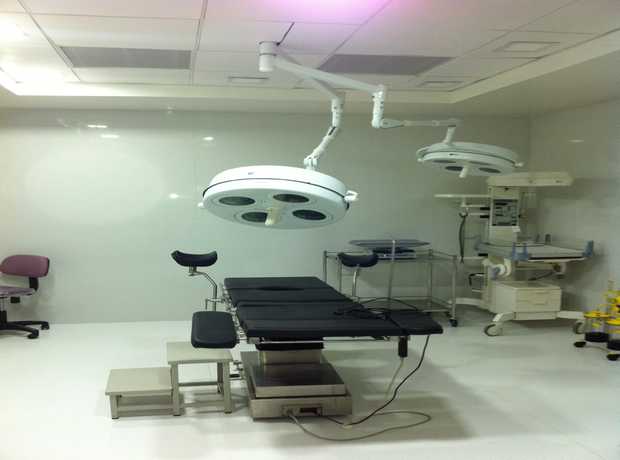
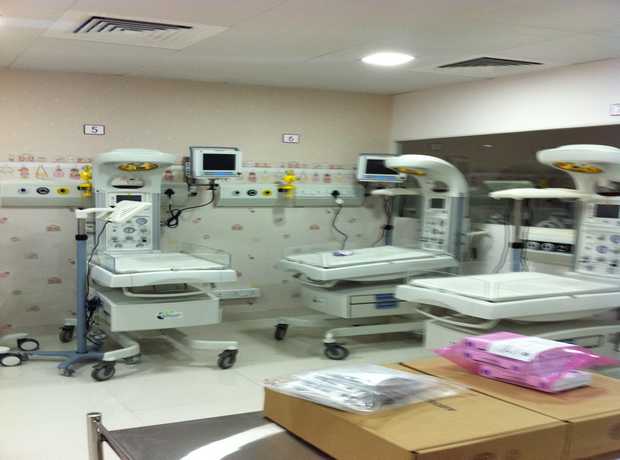
Doha, the vibrant heart of Qatar, is experiencing unprecedented growth, particularly in its healthcare sector. As new hospitals, clinics, and medical facilities emerge to meet the escalating demands of a thriving population, the importance of robust, compliant, and state-of-the-art infrastructure becomes paramount. Within this critical framework, the design and construction of rapid sterilization and autoclave rooms stand out as a cornerstone of patient safety and operational efficiency. This comprehensive guide provides meticulously detailed checklists for the planning, design, execution, and validation of these vital spaces, ensuring adherence to stringent global standards while rigorously conforming to local Doha regulations in 2025 and beyond.
At Skydome Designs, we understand that constructing a sterilization room is not merely a building project; it’s an intricate endeavor demanding specialized expertise, a deep understanding of infection control, and an unwavering commitment to quality. Our nearly three decades of experience, including significant projects across Doha, positions us as your ideal partner in navigating the complexities of this critical build.
Why is Rapid Sterilization and Autoclave Room Construction Critical in Doha’s Evolving Healthcare Landscape?
The role of sterilization rooms, often referred to as Central Sterile Supply Departments (CSSD) or Sterile Processing Departments (SPD), is foundational to preventing healthcare-associated infections (HAIs). These facilities are the unsung heroes of patient safety, responsible for cleaning, disinfecting, sterilizing, and storing reusable medical devices and surgical instruments. In a rapidly expanding healthcare market like Doha, where facilities aim for international accreditation and excellence, having world-class sterilization infrastructure is not just a regulatory requirement; it’s a moral imperative and a competitive advantage.
The specific demands for rapid sterilization and autoclave room construction in Doha stem from several factors:
- Patient Safety: The primary objective is to eliminate pathogens from medical instruments, thereby protecting patients from infections during surgical procedures or other medical interventions.
- Infection Control: A well-designed sterilization room minimizes the risk of cross-contamination between dirty and clean items, safeguarding both patients and healthcare workers.
- Operational Efficiency: Rapid sterilization processes ensure a quick turnaround of instruments, supporting busy operating theatres and emergency departments, crucial for the high-paced environment of modern healthcare.
- Regulatory Compliance: Doha’s Ministry of Public Health (MoPH) and other local authorities enforce strict guidelines that must be met to ensure licensing and operational legality.
- Reputation and Trust: Facilities with superior sterilization capabilities build trust within the community and enhance their reputation for delivering high-quality care.
- Healthcare Expansion: As Doha continues to invest heavily in its healthcare sector, new facilities require robust sterilization units from inception, while existing ones may need upgrades to meet modern standards.
Skydome Designs, with a track record of delivering 1926+ rapid sterilization and autoclave room construction assignments across Doha and globally over 30+ years, consistently achieves 99% on-time delivery. Our multi-disciplinary reviews and post-occupancy support underscore our commitment to optimal outcomes. This extensive experience ensures that your project benefits from proven methodologies and unparalleled expertise.
Understanding the Regulatory Landscape for Autoclave Room Construction in Doha (2025)
Navigating the regulatory environment in Doha is a critical first step for any healthcare construction project. For rapid sterilization and autoclave room construction, this involves a confluence of local Qatari laws and international best practices. Owners, architects, and contractors must be thoroughly conversant with these guidelines to avoid costly delays and ensure the long-term viability and safety of the facility.
Key Regulatory Bodies and Standards:
- Doha Municipality Regulations: These cover general building codes, structural integrity, fire safety, and environmental impact assessments relevant to any construction in the city.
- Ministry of Public Health (MoPH) Guidelines: The MoPH is the primary authority for healthcare facility standards in Qatar. Their guidelines detail specific requirements for infection control, medical equipment, patient safety, and operational protocols within healthcare settings, directly impacting the design and function of sterilization rooms.
- Qatar Civil Defense Department (QCDD): Crucial for fire safety, emergency exits, alarm systems, and ventilation specifications, especially vital for rooms containing high-pressure steam equipment like autoclaves.
- International Standards: Adherence to internationally recognized standards is not only a mark of quality but often a prerequisite for accreditation. Key standards include:
- ISO 15883: Series of standards for washer-disinfectors, covering general requirements, performance, and specific types of equipment.
- ISO 13485: Quality management systems for medical devices, relevant to the processes and environment where devices are handled.
- EN 285: European standard for large steam sterilizers.
- AAMI ST79: Comprehensive guide to steam sterilization and sterility assurance in healthcare facilities (Association for the Advancement of Medical Instrumentation).
- ANSI/ASHRAE/ASHE Standard 170: Ventilation of Health Care Facilities, dictating air changes, pressure relationships, and filtration.
- Local Electrical and Plumbing Codes: Ensuring safety and functionality of all utilities, especially given the heavy power and water demands of sterilization equipment.
Staying abreast of these dynamic regulations and ensuring proactive compliance is a hallmark of successful rapid sterilization and autoclave room construction in Doha. Skydome Designs provides global design standards with local code expertise in Doha, offering end-to-end delivery for rapid sterilization and autoclave room construction—from strategy and design to construction and handover.
The Comprehensive Checklists: A Detailed Roadmap for Success
These meticulously structured checklists offer a phased approach, ensuring that every aspect of your rapid sterilization and autoclave room construction in Doha is covered, from initial concept to final operation.
Phase 1: Planning and Design – Laying the Foundation for Excellence
This initial phase is where critical decisions are made that will dictate the functionality, efficiency, and compliance of the entire sterilization facility. Thorough planning here saves significant time and resources later.
- 1.1 Needs Assessment & Feasibility Study:
- Define the scope and projected capacity based on the healthcare facility’s type (e.g., general hospital, specialized clinic), size, and patient load.
- Estimate the volume and types of instruments to be processed daily, weekly, and monthly.
- Consider future expansion plans and potential increases in service demand; design for modularity or scalability.
- Analyze workflow requirements from instrument reception to sterile storage and distribution.
- Conduct a preliminary site assessment to identify structural, utility, and logistical constraints.
- 1.2 Regulatory Compliance & Accreditation Planning:
- Identify all applicable Doha Municipality regulations, Ministry of Public Health guidelines, Qatar Civil Defense Department requirements, and local fire codes.
- Ensure adherence to relevant international standards such as ISO 15883, EN 285, AAMI ST79, and ASHRAE 170.
- Plan for necessary permits and approvals from all pertinent Qatari authorities well in advance.
- Factor in requirements for potential international accreditations (e.g., JCI) from the outset.
- 1.3 Space Planning & Workflow Optimization:
- Design a clear, unidirectional workflow to prevent cross-contamination, typically following a “dirty-to-clean” path. This includes dedicated zones for:
- Receiving/Decontamination: Where soiled instruments are collected and initially cleaned.
- Preparation & Packaging: Where instruments are inspected, assembled into sets, and packaged for sterilization.
- Sterilization: Housing autoclaves and other sterilizers.
- Sterile Storage: For sterilized items awaiting distribution.
- Administration/Support: Offices, staff changing rooms, utility areas.
- Optimize spatial relationships between zones using visual and physical barriers, such as pass-through windows or cabinets, to maintain segregation.
- Ensure adequate circulation space for personnel and equipment, promoting ergonomic movement and reducing fatigue.
- Plan for appropriate entry and exit points to maintain clean and dirty segregation.
- Design a clear, unidirectional workflow to prevent cross-contamination, typically following a “dirty-to-clean” path. This includes dedicated zones for:
- 1.4 Equipment Selection & Integration:
- Choose autoclaves (steam sterilizers), washer-disinfectors, ultrasonic cleaners, drying cabinets, and other sterilization equipment based on capacity, efficiency, instrument compatibility, and lifecycle costs.
- Prioritize equipment that meets current and future performance criteria, considering energy efficiency and water conservation features for sustainability.
- Evaluate vendor reputation, local service support in Doha, spare parts availability, and warranty conditions.
- Consider sustainability and digital delivery; future-ready rapid sterilization and autoclave room construction lowers lifecycle costs through energy-efficient machines and integrated digital monitoring systems.
- Integrate smart technologies and IoT solutions for real-time monitoring, predictive maintenance, and data logging.
- 1.5 BIM (Building Information Modeling) Integration:
- Implement BIM for accurate 3D design, visualization, and detailed clash detection among architectural, structural, and MEP elements.
- Use BIM for efficient project management, cost estimation, scheduling, and facility lifecycle management from design to operation and maintenance.
- Enhance coordination among multidisciplinary teams, reducing errors and rework during construction.
- 1.6 Budgeting and Cost Management:
- Develop a comprehensive budget covering design, permits, construction, equipment procurement, installation, commissioning, validation, and training.
- Include contingency funds for unforeseen circumstances.
- Analyze the return on investment (ROI) and total cost of ownership, considering operational efficiency and energy consumption.
- 1.7 Risk Assessment and Mitigation:
- Identify potential risks related to design, construction, equipment failure, and operational safety.
- Develop mitigation strategies, including contingency plans for power outages, equipment malfunctions, and supply chain disruptions.
Phase 2: Construction and MEP Services – Precision and Durability
The construction phase translates the design into a physical reality, requiring meticulous attention to detail, adherence to specifications, and seamless integration of complex systems.
- 2.1 Material Selection & Finishes:
- Utilize materials that are durable, non-porous, smooth, easy to clean, and resistant to chemicals, high temperatures, moisture, and frequent disinfection.
- Specify non-shedding, anti-microbial, and anti-static materials for flooring (e.g., seamless epoxy, vinyl), walls (e.g., medical-grade paint, PVC wall protection), and ceilings (e.g., cleanroom-rated tiles).
- Ensure countertops and work surfaces are made of solid, non-porous materials like stainless steel or specialized composites that can withstand rigorous cleaning protocols.
- Doors should be self-closing, wide enough for equipment, and made of durable, cleanable materials.
- 2.2 MEP (Mechanical, Electrical, and Plumbing) Design & Installation:
- HVAC Systems: Design specialized HVAC systems to maintain proper temperature (e.g., 20-23°C), humidity (e.g., 30-60%), and air pressure differentials between zones (e.g., positive pressure in sterile storage, negative pressure in decontamination).
- Ensure a minimum of 6-10 air changes per hour (ACH) in various zones, with appropriate filtration (e.g., HEPA filters for sterile storage) to maintain air quality and minimize airborne contaminants.
- Electrical Systems: Provide robust and dedicated electrical circuits for all equipment, including emergency power backups (UPS/generators) to ensure uninterrupted operation, especially for autoclaves and control systems.
- Ensure adequate lighting levels (e.g., 500-1000 lux) for detailed inspection tasks in preparation and packaging areas, with provisions for dimmable or task lighting.
- Plumbing & Water Systems: Install high-quality plumbing for continuous supply of purified water (e.g., RO/DI water for washer-disinfectors and sterilizers) and efficient drainage systems with floor drains in decontamination areas.
- Ensure steam generators and associated piping are designed for consistent high-quality steam required for sterilization processes.
- Implement backflow prevention devices to protect the potable water supply.
- 2.3 Structural Engineering & Load Bearing:
- Ensure the structural integrity of the floor and building can support the significant weight of heavy equipment such as large autoclaves, washer-disinfectors, and water purification units.
- Design for vibration control, especially for high-speed equipment, to prevent structural damage and operational disruptions.
- Reinforce walls and ceilings where necessary to support suspended equipment, shelving, or utility lines.
- 2.4 Infection Control During Construction:
- Implement strict dust control measures, including temporary barriers, negative pressure zones, and regular cleaning, to prevent construction dust from contaminating existing operational areas of the facility.
- Establish clear pathways for construction personnel and materials, separate from patient and staff traffic flows.
- Enforce strict hygiene protocols for construction workers, including appropriate personal protective equipment (PPE).
- Manage construction waste efficiently to prevent accumulation and potential hazards.
- 2.5 Safety Protocols & Site Management:
- Adhere to all local Qatari safety regulations and international construction safety standards.
- Implement comprehensive safety plans, including fall protection, electrical safety, and confined space entry procedures.
- Conduct regular site inspections and safety briefings.
- 2.6 Project Scheduling and Milestones:
- Develop a detailed project schedule with clear milestones for each phase, utilizing project management tools for tracking progress.
- Ensure effective communication channels are established between all stakeholders: client, architects, engineers, contractors, and equipment suppliers.
Phase 3: Commissioning, Validation, and Handover – Ensuring Operational Readiness
This final phase is crucial for verifying that the sterilization room and its equipment function as intended, are safe for use, and consistently meet required performance standards.
- 3.1 Equipment Installation & Calibration:
- Oversee the professional installation of all sterilization equipment according to manufacturer specifications and local codes.
- Ensure all instruments and controls are precisely calibrated for accurate readings and operation.
- 3.2 Equipment Testing (FAT/SAT):
- Conduct Factory Acceptance Tests (FAT) at the manufacturer’s site and Site Acceptance Tests (SAT) upon installation in Doha to verify proper functionality, safety features, and performance specifications.
- Test all systems, including emergency stops, alarms, and interlocks.
- 3.3 Utilities Testing:
- Perform thorough testing of all MEP systems:
- Air Quality Testing: Verify air changes per hour, differential pressures between zones, particle counts, and HEPA filter integrity.
- Water Quality Testing: Ensure purified water meets specified standards for conductivity, pH, and absence of impurities critical for instrument processing.
- Steam Quality Testing: Verify that steam delivered to autoclaves meets saturation, non-condensable gas, and superheat requirements to ensure effective sterilization.
- Electrical Load Testing: Confirm all electrical systems can handle the operational load and identify any potential weaknesses.
- Perform thorough testing of all MEP systems:
- 3.4 Process Validation (IQ, OQ, PQ):
- Installation Qualification (IQ): Document that the equipment is installed correctly and meets specified requirements.
- Operational Qualification (OQ): Verify that the equipment operates consistently within defined parameters across its operating range.
- Performance Qualification (PQ): Demonstrate that the sterilization process consistently achieves the desired results (e.g., sterility assurance level, SAL of 10-6) under actual use conditions. This involves using biological indicators, chemical indicators, and process challenge devices.
- Validate all sterilization cycles (e.g., gravity, pre-vacuum, flash) for different types of instruments and load configurations.
- 3.5 Documentation and Record Keeping:
- Compile a comprehensive set of “as-built” drawings, equipment manuals, maintenance schedules, testing reports, and validation protocols.
- Create standard operating procedures (SOPs) for all processes within the sterilization room, including equipment operation, cleaning, packaging, sterilization, and quality control.
- Establish a system for ongoing record-keeping of sterilization cycles, equipment maintenance, and quality checks.
- 3.6 Staff Training:
- Provide comprehensive, hands-on training for all staff on equipment operation, maintenance, troubleshooting, and safety procedures.
- Train personnel on infection control protocols, instrument handling, packaging techniques, and documentation requirements.
- Ensure staff are proficient in emergency procedures, including response to equipment failures or power outages.
- 3.7 Maintenance Protocols:
- Establish preventive maintenance schedules for all equipment and facility systems to ensure long-term reliability and performance.
- Ensure access to necessary spare parts and service contracts with equipment suppliers in Doha.
- 3.8 Post-Occupancy Support:
- Plan for post-occupancy reviews and ongoing support to address any issues that arise after the facility becomes operational.
- Establish clear channels for warranty claims and technical assistance.
The Role of Advanced Technologies in Modern Sterilization Room Construction
Modern rapid sterilization and autoclave room construction in Doha is increasingly leveraging cutting-edge technologies to enhance efficiency, safety, and compliance. Integrating these innovations from the design phase ensures a future-ready facility.
- Automation and Robotics: Automated transport systems for instruments, robotic handling for hazardous tasks, and automated packaging systems reduce human error and improve throughput.
- IoT (Internet of Things) and Smart Monitoring: Sensors embedded in equipment and the environment can monitor temperature, humidity, pressure, and equipment status in real-time. This data can be accessed remotely, trigger alerts for deviations, and facilitate predictive maintenance.
- Digital Tracking Systems: Barcoding or RFID tagging of instruments and sets enables complete traceability from patient use to reprocessing and back. This is crucial for infection control investigations and inventory management.
- Advanced Water Purification: State-of-the-art reverse osmosis (RO) and deionization (DI) systems ensure consistently high-purity water, essential for preventing instrument corrosion and biofilm formation.
- Energy-Efficient Autoclaves: Newer models are designed to consume less water and energy, contributing to sustainability goals and reducing operational costs.
- HVAC Controls with AI: Intelligent HVAC systems can dynamically adjust air pressure, temperature, and humidity based on real-time sensor data and occupancy, optimizing environmental conditions while conserving energy.
Environmental Sustainability in Sterilization Room Design for Doha
As Doha commits to sustainable development, integrating eco-friendly practices into rapid sterilization and autoclave room construction is becoming increasingly important. Sustainable design not only benefits the environment but also leads to long-term cost savings for healthcare facilities.
- Energy Efficiency:
- Utilize high-efficiency HVAC systems with heat recovery units.
- Specify LED lighting with motion sensors and daylight harvesting controls.
- Choose autoclaves and washer-disinfectors with low energy consumption and quick cycle times.
- Optimize building envelope design for insulation and thermal performance.
- Water Conservation:
- Install water-efficient fixtures and equipment.
- Implement water recycling systems where feasible, particularly for non-critical processes.
- Monitor water consumption to identify and address inefficiencies.
- Waste Reduction & Management:
- Design for segregation of different waste streams (e.g., general, hazardous, sharps).
- Utilize durable, reusable materials where appropriate to minimize construction waste.
- Consider systems for responsible disposal or recycling of sterilization by-products.
- Indoor Environmental Quality:
- Select low-VOC (Volatile Organic Compound) paints, adhesives, and sealants to improve indoor air quality for staff.
- Ensure adequate ventilation rates and filtration to remove airborne contaminants and maintain a healthy working environment.
Finding the Right Doha Construction Experts for Your Sterilization Project
The complexity and critical nature of rapid sterilization and autoclave room construction in Doha demand a partnership with highly specialized and experienced professionals. Choosing the right firm is paramount to achieving a compliant, efficient, and future-ready facility.
Key Criteria for Selecting Your Construction Partner:
- Proven Track Record in Healthcare Construction: Look for firms with extensive experience specifically in designing and building healthcare facilities, particularly sterile processing departments.
- Expertise in Local Doha Regulations: The partner must have an intimate understanding of Doha Municipality regulations, Ministry of Public Health guidelines, and other Qatari codes.
- Adherence to Global Standards: Ensure the firm is conversant with international benchmarks like ISO 15883, AAMI ST79, and ASHRAE 170.
- BIM Proficiency: A firm that effectively utilizes BIM will offer better coordination, fewer clashes, and a more efficient project delivery.
- MEP and Structural Engineering Acumen: Specialized knowledge in designing robust and compliant MEP systems and structural supports for heavy sterilization equipment is non-negotiable.
- Comprehensive Service Offering: Ideally, the partner should offer end-to-end solutions, from conceptual design to commissioning and handover, streamlining the project.
- Strong Project Management Capabilities: Evidence of disciplined project management, including clear communication, proactive risk management, and adherence to timelines and budgets.
- References and Testimonials: Request references from previous healthcare clients in Doha or similar environments.
Skydome Designs: Your Unrivaled Partner in Doha for Healthcare Interiors and Rapid Sterilization Rooms
At Skydome Designs, we pride ourselves on being more than just a design and construction firm; we are strategic partners in advancing healthcare infrastructure in Doha. With nearly 30 years of experience across India and abroad, our expertise in healthcare interiors is unparalleled, making us the ideal choice for your critical rapid sterilization and autoclave room construction in Doha.
We offer global design standards with local code expertise in Doha. Our approach encompasses end-to-end delivery for rapid sterilization and autoclave room construction — from strategy development and innovative design to efficient construction and seamless handover in Doha. Our integrated team ensures every project is executed with precision, compliance, and an unwavering focus on quality.
Skydome Designs specializes in:
- Hospital Interior Design: From patient-centric rooms, high-tech ICUs, and sterile operating theatres to advanced laboratories and welcoming consultation areas, we meticulously plan and design facility layouts for optimized care delivery.
- Interior Solutions: Our services span comprehensive space planning, ergonomic furniture layouts, cutting-edge lighting design, and full turnkey interior execution, ensuring functional, aesthetic, and compliant spaces.
Why Choose Us for Your Next Project in Doha:
- 29+ years of extensive experience across India and abroad, with a deep understanding of diverse regulatory environments.
- An in-house, multi-disciplinary team of seasoned architects, healthcare planners, interior designers, and project managers, ensuring cohesive and expert delivery.
- A portfolio of award-winning, client-focused, and sustainable designs that consistently exceed expectations.
- A sterling reputation for projects delivered on-time, on-budget, and to the most rigorous global standards.
- We have successfully delivered 1926+ rapid sterilization and autoclave room construction assignments across Doha and globally over 30+ years. Our commitment to excellence is reflected in our 99% on-time delivery rate, complemented by rigorous multi-disciplinary reviews and comprehensive post-occupancy support that underpin superior outcomes.
Contact us today to discuss your rapid sterilization and autoclave room construction project in Doha! 📞 +91 7299072144 | ✉️ info@skydomedesigns.com
Project Management and Quality Assurance: Beyond the Checklists
While checklists provide a structured framework, successful rapid sterilization and autoclave room construction in Doha requires dynamic and robust project management coupled with an unyielding commitment to quality assurance. This holistic approach ensures that the project not only meets technical specifications but also aligns with strategic objectives.
- Robust Project Management:
- Implement agile project methodologies, allowing for flexibility and rapid adaptation to unforeseen challenges.
- Establish clear communication protocols with all stakeholders, including regular site meetings, progress reports, and transparent decision-making processes.
- Proactive risk management, identifying potential issues early and developing mitigation strategies to prevent delays or cost overruns.
- Effective resource allocation, ensuring skilled labor, materials, and equipment are available when needed.
- Strict adherence to schedules and budgets, with continuous monitoring and adjustment as necessary.
- Comprehensive Quality Assurance (QA) Processes:
- Develop a detailed Quality Assurance Plan (QAP) at the project’s outset, outlining standards, inspection points, and responsibilities.
- Implement systematic inspections and quality control checks at every stage, from material procurement to installation and finishing.
- Regular audits to ensure compliance with design specifications, regulatory requirements, and international best practices.
- Utilize independent third-party verification for critical components and processes, adding an extra layer of scrutiny.
- Documentation of all QA activities, including inspection reports, non-conformance reports, and corrective actions taken.
- Stakeholder Engagement:
- Foster a collaborative environment where all team members, including the client, contractors, and suppliers, are engaged and informed.
- Regular feedback loops to address concerns and incorporate client input throughout the project lifecycle.
At Skydome Designs, our commitment to project management excellence is demonstrated through our proven track record. We have successfully delivered 1926+ rapid sterilization and autoclave room construction assignments across Doha and globally over 30+ years. Our 99% on-time delivery rate is a testament to our meticulous planning, multi-disciplinary reviews, and dedicated post-occupancy support, all of which underpin successful outcomes for our clients. Contact us to see how we can ensure success for your next project, guaranteeing a facility that stands as a benchmark for quality and efficiency.
Conclusion
The successful development of rapid sterilization and autoclave room construction in Doha is a complex yet profoundly rewarding undertaking. It demands meticulous planning, unwavering adherence to local and international regulations, and a collaborative partnership with experienced professionals. By diligently following these comprehensive checklists, from initial needs assessment and design to precision construction and rigorous validation, you can ensure your healthcare facility not only meets but exceeds the highest standards of safety, operational performance, and compliance.
Investing in a high-quality sterilization facility is an investment in patient safety, operational efficiency, and the long-term reputation of your healthcare institution in Doha. With its growing healthcare sector, the demand for world-class infrastructure is only set to intensify, making compliant and expertly constructed sterilization rooms an absolute necessity.
Ready to ensure your rapid sterilization and autoclave room project in Doha meets the highest standards? Contact Skydome Designs for expert consultation and unparalleled project management. Our team is ready to transform your vision into a state-of-the-art reality. 📞 +91 7299072144 | ✉️ info@skydomedesigns.com
Frequently Asked Questions (FAQs) About Rapid Sterilization and Autoclave Room Construction in Doha
Here are some common questions and detailed answers regarding the construction of rapid sterilization and autoclave rooms in Doha:
1. What are the key regulations for autoclave room construction in Doha?
Key regulations for autoclave room construction in Doha include a combination of local Qatari laws and international standards. These primarily involve Doha Municipality regulations for general building codes and safety, Ministry of Public Health (MoPH) guidelines for healthcare-specific requirements (especially infection control and medical equipment), Qatar Civil Defense Department (QCDD) requirements for fire safety, and international standards such as ISO 15883 (for washer-disinfectors), EN 285 (for steam sterilizers), AAMI ST79 (comprehensive guide to sterilization), and ASHRAE 170 (for healthcare facility ventilation). Adherence to these is critical for licensing and operational compliance.
2. How can I ensure efficient workflow in a sterilization room?
Efficient workflow in a sterilization room is achieved through meticulous space planning and adherence to the “dirty-to-clean” concept. This involves creating distinct, segregated zones for receiving/decontamination, preparation/packaging, sterilization, and sterile storage. Utilizing pass-through windows or cabinets between zones, strategically placing equipment to minimize movement and cross-contamination, and ensuring adequate circulation space for staff and trolleys are crucial design elements. An ergonomic layout that reduces unnecessary steps and promotes smooth progression of instruments enhances both efficiency and safety.
3. What are the essential MEP (Mechanical, Electrical, and Plumbing) considerations for autoclave rooms?
Essential MEP considerations for autoclave rooms are complex due to the specialized nature of the equipment. These include:
- HVAC: Designing robust HVAC systems to maintain precise temperature (20-23°C), humidity (30-60%), and critical air pressure differentials (e.g., negative in decontamination, positive in sterile storage) with appropriate air changes per hour (6-10 ACH) and HEPA filtration in sterile areas.
- Electrical: Providing dedicated, heavy-duty electrical circuits with sufficient capacity for high-power equipment, along with reliable emergency power backups (UPS/generators) to prevent cycle interruptions.
- Plumbing: Ensuring a continuous supply of high-purity water (RO/DI water) for washer-disinfectors and sterilizers, robust drainage systems with floor drains in wet areas, and a reliable source of high-quality steam for autoclaves.
4. Why is BIM important for autoclave room construction?
Building Information Modeling (BIM) is invaluable for autoclave room construction because it allows for the creation of a comprehensive 3D digital model of the facility. This enables accurate design visualization, early detection and resolution of clashes between architectural, structural, and MEP systems, and efficient project management from conception to completion. BIM facilitates better collaboration among all stakeholders, improves cost estimation, optimizes scheduling, reduces errors, minimizes rework on-site, and supports long-term facility management, ultimately leading to a more streamlined and successful project delivery.
5. How do I choose the right equipment for my sterilization room?
Choosing the right equipment for your sterilization room requires a detailed assessment of several factors:
- Capacity: Determine the volume of instruments and medical devices your facility will process daily.
- Types of Materials: Consider the specific types of instruments and materials (e.g., heat-sensitive, non-heat-sensitive) to ensure compatibility with chosen sterilization methods (e.g., steam, low-temperature sterilization).
- Performance Criteria: Select equipment that meets high performance, reliability, and safety standards.
- Sustainability: Evaluate energy and water efficiency, digital integration capabilities, and automation features for future readiness.
- Vendor Support: Prioritize manufacturers and suppliers with excellent local service, maintenance, and spare parts availability in Doha.
- Budget & Lifecycle Cost: Balance initial purchase costs with long-term operational and maintenance expenses.
6. What are the key considerations for infection control during the construction of a sterilization room?
Infection control during construction is paramount, especially when building within or adjacent to an operational healthcare facility. Key considerations include:
- Containment: Erecting robust physical barriers and implementing negative air pressure zones to prevent the spread of dust, debris, and contaminants into clean areas.
- Dust Control: Implementing strict measures like dampening surfaces, using HEPA-filtered vacuums, and installing temporary air scrubbers.
- Worker Hygiene: Enforcing strict protocols for construction personnel, including wearing appropriate PPE, dedicated entry/exit points, and hand hygiene.
- Material Handling: Ensuring construction materials are stored and transported in a way that minimizes contamination risks.
- Waste Management: Implementing an efficient and safe waste disposal system for construction debris.
For a free consultation on your rapid sterilization and autoclave room construction project in Doha, contact Skydome Designs, your trusted partner for excellence in healthcare infrastructure. 📞 +91 7299072144 | ✉️ info@skydomedesigns.com
Skydome Designs Pvt Ltd is a leading architecture and interior design firm in India, specializing in hospital and healthcare interiors, residential, and retail projects. With nearly 30 years of expertise, we deliver innovative, sustainable, and functional spaces that enhance experiences and operational efficiency across the globe, including significant contributions to Doha’s evolving healthcare landscape.