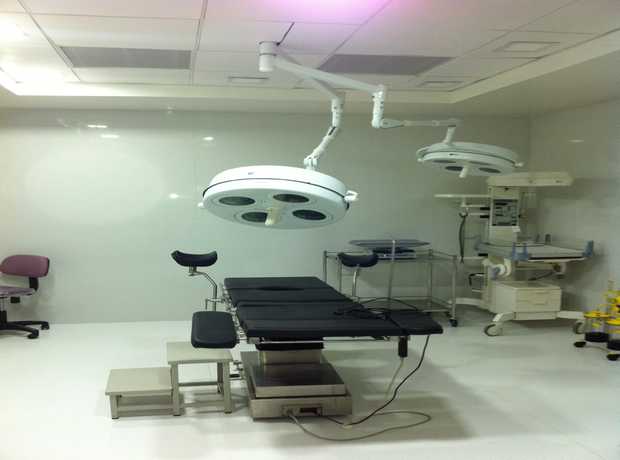
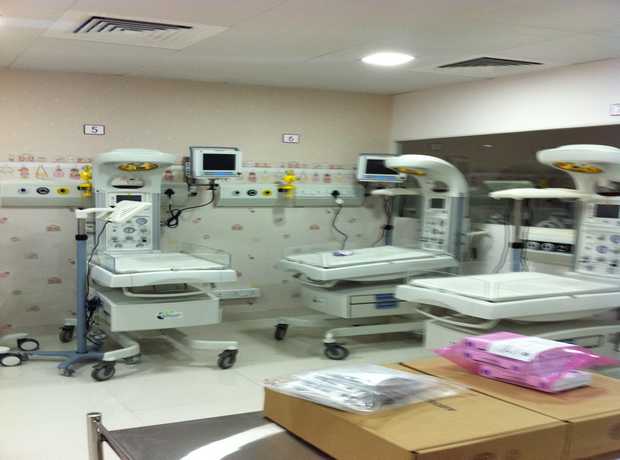
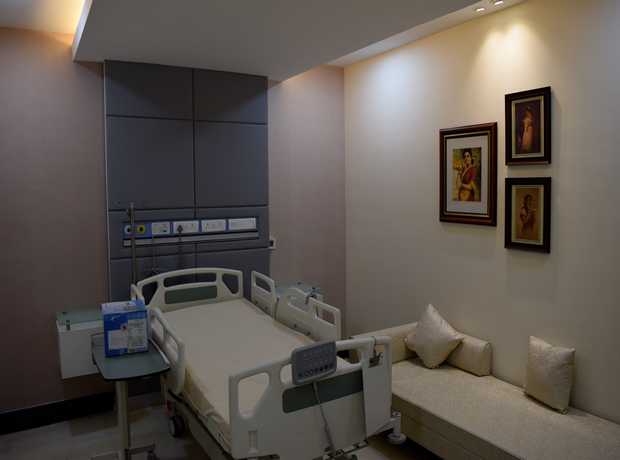
The pharmaceutical industry in Dubai is experiencing significant, rapid growth, fueled by strategic government initiatives, a burgeoning healthcare sector, and its position as a global trade hub. This expansion demands not just more facilities, but state-of-the-art infrastructure that meets and often exceeds stringent global manufacturing and quality assurance standards. Planning and executing pharmaceutical facility interiors for Dubai projects in 2025 and beyond requires an incredibly meticulous, multi-disciplinary approach that integrates cutting-edge technology, robust regulatory adherence, and sustainable practices.
This comprehensive guide delves deep into each critical step of designing, constructing, and validating a pharmaceutical facility in Dubai, ensuring your project’s success from conception to commissioning. With Dubai continuously prioritizing innovation, digital transformation, and sustainability, future-ready pharmaceutical facility interiors are not just a luxury but a necessity, significantly lowering lifecycle costs and enhancing operational resilience. Partnering with experts like Skydome Designs, who bring a wealth of experience and a proven track record, is pivotal in navigating the complexities of this specialized sector.
1. Initial Consultation and Strategic Project Definition for Dubai Pharmaceutical Facilities
The foundational phase of any successful pharmaceutical project in Dubai begins with an in-depth, detailed consultation. This is where your vision is translated into a clear, actionable project brief. Understanding your specific needs and objectives is paramount, as it dictates every subsequent design and construction decision. This step is not merely about identifying requirements; it’s about strategic alignment with market demands, regulatory expectations, and your long-term business goals.
- Understanding Your Needs: Defining the Facility’s Purpose: This involves clearly articulating the type of pharmaceutical facility required. Will it be a manufacturing plant for sterile injectables, oral solids, or biologics? Is it a cutting-edge research and development (R&D) laboratory, a specialized quality control (QC) laboratory, or a high-capacity storage and distribution center? Each type has unique requirements impacting layout, material specifications, environmental controls, and regulatory pathways. We delve into process flows, production volumes, future expansion plans, and specific technology integration needs.
- Navigating Regulatory Compliance: The Dubai & Global Landscape: Dubai operates within a robust regulatory framework that aligns with international best practices. Identifying all applicable local and international regulations is crucial from day one. This includes, but is not limited to, Dubai Municipality regulations for building codes and environmental standards, the Dubai Health Authority (DHA) for licensing and operational guidelines, and the Ministry of Health and Prevention (MOHAP). Crucially, compliance extends to global benchmarks such as Good Manufacturing Practices (GMP) as defined by organizations like the World Health Organization (WHO GMP), Pharmaceutical Inspection Co-operation Scheme (PIC/S GMP), US FDA, and the European Medicines Agency (EMA). Understanding these nuanced requirements ensures that the facility is built to global standards, enabling easier market access and product registration.
- Establishing a Realistic Budget and Project Timeline: Financial and temporal planning are critical pillars. This involves establishing a detailed budget that accounts for design, construction, equipment, validation, and contingency. Similarly, a realistic project timeline, broken down into key milestones, is developed to manage expectations and ensure timely delivery. Given Dubai’s dynamic business environment, incorporating flexibility for unforeseen challenges while maintaining strict control over resources is key.
Considering Dubai’s unwavering focus on innovation and digital transformation, it’s not enough to just meet standards; it’s crucial to integrate advanced digital solutions and sustainable practices from the outset. This forward-thinking approach future-proofs your investment and aligns with the Emirate’s strategic vision. For an initial consultation that lays the groundwork for your project’s success, contact Skydome Designs at +91 7299072144 or info@skydomedesigns.com to schedule your initial consultation. Our team is ready to transform your vision into a compliant, efficient, and sustainable reality.
2. Conceptual Design and Strategic Space Planning for Optimal Pharmaceutical Operations
Once the project’s foundational requirements are clear, the conceptual design phase commences, translating initial ideas into tangible spatial configurations. This phase is less about aesthetics and more about intelligent functionality, workflow optimization, and ensuring absolute adherence to safety and quality standards. Skydome Designs excels in this critical stage, meticulously crafting functional yet aesthetically pleasing spaces that prioritize maximum operational efficiency, stringent compliance, and enhanced user experience.
- Workflow Analysis and Lean Design Principles: A pharmaceutical facility’s efficiency is directly tied to its workflow. Our approach involves a rigorous analysis of material flow (raw materials to finished products) and personnel flow (from entry to specific work zones). The goal is to design a layout that minimizes cross-contamination, reduces travel distances, eliminates bottlenecks, and optimizes sequential processes. This often involves applying lean manufacturing principles to streamline operations, reduce waste, and improve overall productivity. Segregation of activities – from dispensing and manufacturing to packaging, quality control, and warehousing – is carefully planned to prevent contamination and ensure product integrity.
- Critical Equipment Integration and Advanced Technology: Pharmaceutical manufacturing relies on highly specialized equipment, each with specific spatial, utility, and environmental needs. This phase involves precise planning for the placement of key equipment, including reactors, blenders, tablet presses, filling lines, and packaging machinery. Crucially, it also encompasses the integration of complex HVAC (Heating, Ventilation, and Air Conditioning) systems, which are central to maintaining cleanroom environments, and specialized cleanroom technology such as air showers, pass boxes, and laminar flow hoods. The design must accommodate utility connections, maintenance access, and future equipment upgrades without disrupting operations.
- Rigorous Safety and Environmental Considerations: Safety is non-negotiable in pharmaceutical facilities. The conceptual design phase incorporates comprehensive safety features, including strategically placed fire suppression systems, clearly marked emergency exits, accessible safety showers, and eye wash stations. Furthermore, provisions for handling hazardous materials, controlled waste management, and spill containment are integrated into the initial layout. Environmental considerations extend to designing spaces that allow for optimal ergonomic conditions for staff, adequate natural light where appropriate, and noise control to create a conducive and safe working environment.
This conceptualization is where the blueprint for sterile environments, controlled zones, and efficient logistics takes shape. Our team, with its deep understanding of pharmaceutical processes, ensures that every square meter is optimized for its intended purpose, setting the stage for a facility that is not only compliant but also highly productive and future-proof. With Skydome Designs, you benefit from our award-winning team’s global design standards combined with local code expertise in Dubai, ensuring a facility that stands out for its excellence and adherence to regional specifics.
3. Detailed Design and Comprehensive Engineering for Pharmaceutical Precision
The detailed design phase is where the conceptual plans are transformed into highly precise, actionable blueprints and specifications. This complex stage brings together various engineering disciplines to create a comprehensive design package that forms the backbone of the construction process. Engaging experienced Dubai construction experts and specialized engineering consultants is absolutely crucial during this phase to ensure technical accuracy, regulatory compliance, and constructability.
3.1 Structural Engineering: Building Resilience in Dubai
Structural integrity is the bedrock of any pharmaceutical facility, especially when considering Dubai’s unique environmental conditions. Our structural engineering designs meticulously account for high temperatures, varying humidity levels, and potential seismic activity, ensuring long-term durability and safety. This involves:
- Material Specification: Selecting robust construction materials that can withstand the demanding operational environment, including specific requirements for vibration control from heavy machinery.
- Load Bearing Capacities: Designing foundations, floors, and support structures to safely accommodate heavy pharmaceutical equipment, utilities, and material storage.
- Fire and Safety Compliance: Integrating fire-rated constructions, evacuation routes, and emergency load paths as per international and Dubai civil defense codes.
- Building Envelope: Ensuring a tightly sealed building envelope to maintain internal environmental controls and energy efficiency, crucial for cleanroom operations.
3.2 MEP (Mechanical, Electrical, Plumbing) Services: The Lifeblood of a Pharma Facility
The MEP systems are arguably the most critical components of a pharmaceutical facility, directly impacting product quality, environmental control, and operational safety. Our designs are tailored for pharmaceutical operations, focusing on efficiency, redundancy, and meticulous control.
- HVAC Systems and Cleanroom Technology: This is the heart of cleanroom integrity. We design highly sophisticated HVAC systems to maintain precise temperature, humidity, and pressure differentials across different cleanroom classifications (e.g., ISO Class 5 to ISO Class 8, or EU GMP Grade A to D). This includes multi-stage filtration (pre-filters, HEPA/ULPA filters), dedicated air handling units (AHUs), intelligent airflow patterns (unidirectional, turbulent), and energy recovery systems. Proper ventilation, air changes per hour (ACH), and containment strategies are paramount to prevent cross-contamination and ensure operator safety.
- Electrical Power Distribution and Redundancy: Uninterrupted power is vital for critical processes. Designs include robust power distribution networks, uninterruptible power supply (UPS) systems for essential equipment, and reliable emergency backup generators. Cleanroom-specific lighting, often indirect or recessed, is chosen to minimize particle generation and facilitate easy cleaning.
- Water Management Systems: Purified Water (PW) and Water for Injection (WFI): Pharmaceutical manufacturing demands ultra-pure water. We design and integrate advanced water purification systems (e.g., reverse osmosis, deionization, distillation) for producing Purified Water (PW) and Water for Injection (WFI), adhering to pharmacopoeial standards. This also includes specialized piping materials (e.g., stainless steel 316L for WFI loops), distribution loops, and comprehensive drainage systems for process wastewater and hazardous waste, ensuring environmental compliance.
- Process Utilities: Beyond standard MEP, pharmaceutical facilities require specialized process utilities such as compressed air (instrument air, plant air), nitrogen (for inerting), vacuum systems, and steam. Each system is designed for purity, reliability, and appropriate distribution to points of use.
- Building Management Systems (BMS) / Building Automation Systems (BAS): Modern pharmaceutical facilities leverage sophisticated BMS/BAS to monitor and control critical parameters in real-time – temperature, humidity, pressure, particle counts, and equipment status. This ensures continuous compliance, optimizes energy consumption, and provides crucial data for quality assurance and facility management.
3.3 BIM (Building Information Modeling) and Advanced Digital Integration
The adoption of BIM has revolutionized construction, and in Dubai, it’s becoming a mandate for major projects. Utilizing BIM goes beyond 3D modeling; it’s a holistic digital representation of the facility’s physical and functional characteristics. Its importance in pharmaceutical facility design is amplified:
- Enhanced Collaboration and Communication: BIM facilitates real-time collaboration among architects, engineers, contractors, and stakeholders, breaking down silos and improving decision-making.
- Clash Detection and Conflict Resolution: Before construction begins, BIM models can identify potential clashes between structural, architectural, and MEP elements, resolving conflicts digitally to avoid costly on-site errors and delays.
- Visualization and Walkthroughs: High-fidelity BIM models allow for virtual walkthroughs, helping clients visualize the space, optimize layouts, and make informed choices about equipment placement and workflow.
- Lifecycle Management and Digital Twins: BIM data can be leveraged throughout the facility’s lifecycle – from design and construction to operations and maintenance. It forms the basis for creating a ‘digital twin’ of the facility, enabling predictive maintenance, energy optimization, and easier future modifications.
- Alignment with Dubai’s Digital Delivery Mandate: Dubai is actively prioritizing digital delivery in construction projects. By embracing BIM, your project aligns with these directives, enhancing efficiency, transparency, and ultimately project success.
Our commitment to excellence in the detailed design and engineering phase is unwavering. Skydome Designs operates with global design standards and local code expertise in Dubai. Our award-winning team ensures transparent costs and milestone-based reporting in Dubai, providing unparalleled clarity and control throughout the project. This meticulous planning ensures that your facility is not just built, but engineered for precision, compliance, and longevity.
4. Strategic Material Selection and Rigorous Procurement for Pharmaceutical Environments
The selection and procurement of materials for pharmaceutical facility interiors are far from standard construction practices. Every material choice has a direct impact on product quality, contamination control, operational efficiency, and long-term maintenance. This phase requires an intimate understanding of pharmacopoeial requirements, cleanroom standards, and Dubai’s climate considerations.
- Hygiene and Cleanliness: The Foremost Criterion: The paramount consideration is the ability of materials to prevent contamination. This means selecting surfaces that are seamless, non-porous, non-shedding, and exceptionally easy to clean and sanitize. Materials must be resistant to common cleaning agents, disinfectants, and potential chemical spills.
- Flooring: Seamless epoxy or polyurethane (PU) self-leveling flooring systems are preferred, often with coved skirting (curved transitions from floor to wall) to eliminate dust-collecting corners. Anti-static properties may also be required in certain zones.
- Walls: Modular cleanroom panels, typically made from high-pressure laminate (HPL), pre-painted galvanized iron (PPGI), or stainless steel, are chosen for their smooth, non-shedding, and easily sanitizable surfaces. Flush-mounted windows and doors are essential.
- Ceilings: Walkable or non-walkable modular cleanroom ceiling systems, often with integrated lighting and HEPA filter terminals, ensure a sealed, clean environment above the production areas.
- Doors and Windows: Hermetically sealed, flush-mounted doors (often automatic or semi-automatic) and double-glazed windows designed to prevent condensation and air leakage are standard.
- Durability and Resistance for Demanding Operations: Pharmaceutical environments are subjected to heavy usage, rigorous cleaning, and exposure to various chemicals. Materials must be selected for their exceptional durability and resistance to wear, abrasion, impact, and chemical degradation. This includes resistance to acids, bases, solvents, and biological agents. The structural integrity of walls and floors must withstand the weight and movement of heavy equipment.
- Sustainability and Environmental Stewardship in Dubai: Aligning with Dubai’s strong commitment to green building initiatives and sustainability goals (such as Estidama or LEED certification) is increasingly important. This involves opting for environmentally friendly materials that have a low carbon footprint, are locally sourced where possible, are recyclable, and contribute to improved indoor air quality. We prioritize materials that offer long-term performance, reducing the need for frequent replacement and minimizing waste over the facility’s lifecycle. Considerations include volatile organic compound (VOC) emissions, energy efficiency of manufacturing processes for materials, and responsible sourcing.
- Procurement Strategy and Supply Chain Management: Sourcing specialized pharmaceutical-grade materials requires a robust procurement strategy. This includes identifying reputable suppliers who can provide certified materials, ensuring timely delivery to meet project schedules, and managing logistics within Dubai’s import/export frameworks. Quality assurance extends to verifying material specifications, certifications, and batch traceability from the supplier.
Every material chosen for your Dubai pharmaceutical facility interior is a deliberate decision aimed at supporting an aseptic environment, ensuring operational longevity, and contributing to the highest standards of product quality and safety. Skydome Designs’ deep expertise in pharmaceutical material science ensures that your facility is built with the right components from the ground up.
5. Rigorous Construction and Meticulous Project Management: Bringing Vision to Reality in Dubai
The construction phase is the culmination of all prior planning and design efforts, where blueprints materialize into physical structures. Executing this phase for a pharmaceutical facility requires exceptional project management, characterized by meticulous coordination, strict adherence to quality control, and unwavering focus on safety and regulatory compliance. It’s a complex dance involving numerous stakeholders, specialized contractors, and critical timelines. Skydome Designs provides comprehensive project management services, honed over decades, ensuring not just on-time and on-budget delivery, but also strict adherence to pharmaceutical industry best practices.
- Site Supervision and Quality Assurance: Our dedicated site supervision teams ensure that every aspect of construction aligns precisely with the approved design specifications, engineering drawings, and quality standards. Regular, rigorous site inspections are conducted to monitor progress, identify potential deviations, and implement corrective actions swiftly. Quality assurance encompasses everything from civil structural integrity to the precise installation of cleanroom panels, HVAC ducts, and utility lines. We employ a proactive approach to quality, preventing issues before they arise, rather than merely inspecting after the fact.
- Implementing Rigorous Quality Control (QC) Measures: Beyond general construction quality, pharmaceutical facilities demand specialized QC. This involves verifying the integrity of cleanroom envelopes, testing the sealing of joints, ensuring proper installation of coved skirtings, and confirming the smooth, non-porous finish of surfaces. All installations, especially for critical systems like HVAC, water purification, and electrical, undergo stringent checks against pharmaceutical benchmarks and approved vendor specifications. Documentation of all QC checks is maintained meticulously, forming part of the overall validation package.
- Effective Communication and Coordination Among Stakeholders: A pharmaceutical construction project involves a vast ecosystem of stakeholders: the client, architects, structural engineers, MEP engineers, cleanroom specialists, equipment suppliers, general contractors, and numerous subcontractors. Our project management methodology emphasizes robust communication channels and seamless coordination. Regular meetings, detailed progress reports, and a centralized information flow ensure that all parties are aligned, potential conflicts are resolved promptly, and project milestones are met without delays. This proactive coordination is crucial for navigating the complexities of integrating specialized pharmaceutical equipment and systems.
- Health, Safety, and Environmental (HSE) Protocols: Construction sites, especially those involving specialized equipment and sensitive environments, demand the highest HSE standards. We implement comprehensive HSE management plans that include regular safety audits, mandatory safety training for all personnel, and strict adherence to local Dubai regulations and international best practices for construction safety. This also covers the safe handling and disposal of construction waste and compliance with environmental protection guidelines.
- Specialized Cleanroom Construction Practices: The construction of cleanrooms involves unique methodologies, such as phased installation, rigorous cleanliness protocols during construction, and careful control over dust and debris generation. Air quality monitoring is often initiated even during construction to prevent contamination of the cleanroom envelope.
Our extensive experience speaks volumes: Skydome Designs has delivered 630+ pharmaceutical facility interiors assignments across Dubai and globally over 12+ years. Our on-time delivery rate of 97%, combined with multi-disciplinary reviews and post-occupancy support, underpins the exceptional outcomes we consistently achieve. Choosing Skydome Designs means partnering with a firm that understands the nuances of pharmaceutical construction and project management in Dubai, ensuring your investment translates into a world-class, fully compliant, and operationally efficient facility. Contact us today to leverage our proven expertise.
6. Meticulous Validation and Precise Commissioning for Operational Readiness
The transition from construction to operational readiness in a pharmaceutical facility is governed by a critical and highly regulated process: validation and commissioning. This phase is non-negotiable, ensuring that every system, piece of equipment, and operational procedure meets stringent regulatory requirements, design specifications, and performance standards. For Dubai projects, adhering to global design standards with local code expertise is paramount to achieve regulatory approval and maintain product quality.
- Equipment Validation (IQ, OQ, PQ): This three-tiered approach ensures that individual pieces of equipment are fit for purpose:
- Installation Qualification (IQ): Verifies that the equipment has been installed correctly, according to manufacturer specifications and design documents. This includes checking utilities, calibration, and confirming proper connections.
- Operational Qualification (OQ): Confirms that the equipment functions as intended across its anticipated operating ranges. This involves testing control limits, alarms, and all operational sequences under various conditions.
- Performance Qualification (PQ): Demonstrates that the equipment consistently performs as expected under actual operating conditions, producing acceptable results over a sustained period, meeting predefined quality attributes.
- System Validation: Ensuring Critical Infrastructure Performance: Beyond individual equipment, entire critical systems must be validated. This includes:
- HVAC Systems: Validation protocols for HVAC verify air changes, temperature and humidity control, pressure differentials between zones, particle counts (for cleanroom classification, e.g., ISO 14644), and filter integrity (DOP/PAO testing for HEPA filters).
- Water Purification Systems: Validation ensures the consistent production of Purified Water (PW) and Water for Injection (WFI) meeting pharmacopoeial monographs, including microbiological and chemical purity tests. This involves extensive sampling and testing of the generation and distribution loops.
- Cleanroom Technology: All cleanroom components, from airlocks and pass boxes to laminar flow units, are validated to ensure they contribute to maintaining the required environmental conditions.
- Process Control Systems: Automation systems, SCADA, and other control systems that manage critical processes are rigorously tested for accuracy, reliability, and data integrity.
- Comprehensive Documentation: The Backbone of Compliance: Every step of the validation and commissioning process generates extensive documentation. This includes:
- Validation Master Plan (VMP): An overarching document outlining the validation strategy for the entire facility.
- Design Qualification (DQ): Verifying that the design itself meets user requirements and regulatory guidelines.
- IQ, OQ, PQ Protocols and Reports: Detailed documents outlining the tests performed, results, deviations, and conclusions for each equipment and system.
- Standard Operating Procedures (SOPs): Development of comprehensive SOPs for facility operation, maintenance, cleaning, and quality control.
This documentation package is crucial for regulatory audits, ensuring traceability, and demonstrating continuous compliance throughout the facility’s operational life.
- Regulatory Audits and Certification: Post-validation, the facility will undergo audits by local (DHA, MOHAP) and potentially international regulatory bodies (e.g., WHO GMP, PIC/S) to grant operational licenses and certifications. Successful validation is the gateway to these approvals.
Our expertise ensures that your pharmaceutical facility in Dubai not only passes these rigorous tests but also operates at peak performance from day one. Skydome Designs combines global design standards with local code expertise in Dubai, providing award-winning, client-focused designs that inherently simplify the validation process. This meticulous approach minimizes risks, accelerates market entry, and assures the highest levels of product quality and patient safety.
7. Smooth Handover and Enduring Post-Occupancy Support
The completion of construction, validation, and commissioning marks a pivotal transition: the handover of the fully operational pharmaceutical facility to the client. However, Skydome Designs’ commitment extends far beyond this point, encompassing comprehensive handover procedures and crucial post-occupancy support to ensure sustained performance and client satisfaction.
- Comprehensive Handover Documentation and Training Programs: The handover process is meticulously managed, providing the client with a complete package of “as-built” drawings, operation and maintenance (O&M) manuals, equipment warranties, and all relevant validation documentation. Crucially, we facilitate detailed training programs for your personnel. This includes hands-on instruction on the operation of complex equipment, the use of critical systems (HVAC, water purification), routine maintenance schedules, emergency procedures, and cleanroom protocols. Empowering your team with the necessary knowledge ensures smooth, compliant, and efficient operation from day one.
- Proactive Maintenance Services and Ongoing Support: A pharmaceutical facility is a living entity that requires continuous care to remain in optimal condition. Skydome Designs offers tailored ongoing maintenance and support services designed to maximize uptime, extend asset life, and ensure continuous regulatory compliance. This can include scheduled preventative maintenance for critical systems, calibration services for instruments, and regular performance checks of the cleanroom environment. Our proactive approach helps identify and address potential issues before they impact operations or product quality.
- Responsive Troubleshooting and Continuous Improvement: Despite meticulous planning, operational challenges can sometimes arise. Our post-occupancy support includes prompt troubleshooting services to address any issues or concerns that may emerge after the facility becomes operational. Furthermore, we believe in a partnership for continuous improvement. As your operations evolve or as new technologies and regulations emerge, we stand ready to assist with facility upgrades, modifications, or process optimizations. This long-term commitment ensures that your facility remains at the forefront of pharmaceutical manufacturing and always meets evolving industry demands.
Our dedication to client success is evident in our track record. Skydome Designs has delivered 630+ pharmaceutical facility interiors assignments across Dubai and globally over 12+ years. Our 97% on-time delivery rate, multi-disciplinary reviews, and comprehensive post-occupancy support are testaments to our unwavering commitment to client outcomes. We view the handover not as an end, but as the beginning of a sustained partnership focused on the enduring success and optimal performance of your pharmaceutical facility in Dubai.
Why Choose Skydome Designs for Your Dubai Pharmaceutical Facility Interiors?
Navigating the intricate landscape of pharmaceutical facility design and construction in Dubai demands a partner with unparalleled expertise, a proven track record, and a forward-thinking approach. Skydome Designs stands as a beacon of excellence in this specialized field, offering distinct advantages that set your project apart.
- 29+ Years of Unmatched Experience Across India and Abroad: Our nearly three decades of experience span a vast portfolio of projects, not only across diverse regions in India but also internationally. This extensive exposure has endowed us with a profound understanding of varied regulatory environments, construction methodologies, and client expectations, making us exceptionally well-equipped to handle the specific challenges and opportunities presented by the Dubai market.
- An In-House Powerhouse of Talent: We pride ourselves on an integrated, in-house team comprising highly skilled architects, specialized healthcare planners (with deep pharma sector knowledge), and experienced project managers. This multi-disciplinary collaboration ensures seamless communication, efficient problem-solving, and a holistic approach to design and delivery. Our experts work in synergy, ensuring that every facet of your pharmaceutical facility – from architectural aesthetics to complex process flows and stringent compliance requirements – is meticulously addressed.
- Award-Winning, Client-Focused, and Sustainable Designs: Our commitment to innovative design has been recognized through numerous awards. Beyond accolades, our design philosophy is deeply client-centric, focusing on understanding your unique operational needs, brand identity, and long-term vision. We are pioneers in integrating sustainable design principles, developing energy-efficient solutions, utilizing environmentally responsible materials, and designing facilities that minimize their ecological footprint while lowering operational costs in the long run. Our designs are not just functional; they are future-ready and responsible.
- Unwavering Commitment to On-Time, On-Budget, Global Standard Delivery: In the fast-paced and competitive pharmaceutical sector, time and cost efficiencies are paramount. Our robust project management methodologies, transparent cost reporting, and milestone-based tracking ensure that your project is delivered precisely on schedule and within the agreed budget. Furthermore, our designs consistently adhere to global pharmaceutical standards (like WHO GMP, PIC/S, US FDA), coupled with an intricate understanding of local Dubai regulations, guaranteeing a facility that is compliant anywhere in the world.
- Proven Track Record in Pharmaceutical Facility Interiors: Our extensive experience specifically in pharmaceutical facility interiors speaks for itself. Skydome Designs has successfully delivered 630+ pharmaceutical facility interiors assignments across Dubai and globally over 12+ years. Our remarkable 97% on-time delivery rate, coupled with multi-disciplinary reviews and comprehensive post-occupancy support, consistently underpins successful outcomes for our clients. This unparalleled experience means we anticipate challenges, implement best practices, and deliver reliable, high-performing facilities.
- Transparent Costs and Milestone-Based Reporting: Financial clarity and accountability are cornerstones of our service. We provide transparent cost breakdowns and detailed milestone-based reporting throughout the project lifecycle. This ensures you have full visibility into your investment and can track progress against agreed benchmarks, fostering trust and confident decision-making.
Choosing Skydome Designs for your Dubai pharmaceutical facility interiors means partnering with a firm that brings nearly 30 years of global expertise, a client-focused ethos, and an unwavering commitment to delivering innovative, sustainable, and functional spaces. Our designs not only enhance operational efficiency but also create environments that foster innovation and support the highest standards of pharmaceutical production. Call us today to discuss how we can bring your vision to life: +91 7299072144 or email info@skydomedesigns.com for a consultation.
Conclusion: Building the Future of Pharma in Dubai with Skydome Designs
Designing and constructing state-of-the-art pharmaceutical facility interiors in Dubai is a monumental undertaking, demanding a structured, meticulous, and expert-driven approach. As Dubai continues its ascent as a global healthcare and innovation hub, the demand for facilities that are not only compliant with the most stringent global standards but also embody sustainability, digital integration, and operational excellence will only intensify.
By following the comprehensive step-by-step guidelines outlined in this guide – from initial consultation and conceptual design to detailed engineering, material selection, rigorous construction, meticulous validation, and robust post-occupancy support – your project can achieve unparalleled success. Each phase is critical, and the interdependencies demand a partner who possesses both deep industry knowledge and localized expertise.
Skydome Designs stands as that indispensable partner. With nearly three decades of experience, an award-winning in-house team, and a proven track record of delivering 630+ pharmaceutical facility interiors assignments across Dubai and globally over 12+ years, we bring the global design standards and local code expertise crucial for navigating the Dubai landscape. Our commitment to transparent costs, milestone-based reporting, a 97% on-time delivery rate, and comprehensive post-occupancy support ensures that your investment yields a facility that is future-ready, compliant, and optimized for peak performance.
Entrusting your pharmaceutical facility project to Skydome Designs means choosing innovation, reliability, and a steadfast commitment to quality. We are dedicated to translating your vision into a high-performing, sustainable asset that contributes significantly to the growth of Dubai’s vibrant pharmaceutical industry. Don’t just build a facility; build a legacy of excellence.
Contact Skydome Designs today to start planning your pharmaceutical facility project and experience the difference that true expertise makes!
Email Us: info@skydomedesigns.com
FAQ: Pharmaceutical Facility Interiors in Dubai
Here are some frequently asked questions about pharmaceutical facility interiors in Dubai, optimized for featured snippets and comprehensive understanding:
What are the absolute key considerations for designing pharmaceutical facility interiors in Dubai?
The absolute key considerations for designing pharmaceutical facility interiors in Dubai encompass strict regulatory compliance (including Dubai Municipality regulations, DHA, MOHAP, and international GMP guidelines like WHO GMP, PIC/S, US FDA), meticulous workflow optimization to prevent cross-contamination, stringent hygiene standards, careful material selection for cleanability and durability, and seamless integration of advanced technologies such as BIM for efficiency and lifecycle management. It is crucial to merge global design standards with local code expertise.
How can I ensure my pharmaceutical facility meets all regulatory requirements in Dubai and globally?
To ensure full regulatory compliance, engage experienced consultants and architects, like Skydome Designs, who are intimately familiar with both Dubai’s local regulatory landscape and international GMP guidelines. Implement a comprehensive Validation Master Plan, conducting thorough Design Qualification (DQ), Installation Qualification (IQ), Operational Qualification (OQ), and Performance Qualification (PQ) processes for all equipment and systems. Maintain meticulous documentation throughout every project phase. Skydome Designs offers end-to-end support to guarantee compliance and successful audits.
What critical role does project management play in pharmaceutical facility construction in Dubai?
Effective project management is paramount in pharmaceutical facility construction in Dubai. It is crucial for expert coordination among diverse contractors, specialized suppliers, and multiple stakeholders, ensuring seamless integration of complex systems. Robust project management ensures projects are delivered precisely on-time, within-budget, and to stringent global quality standards. Key aspects include rigorous quality control, proactive risk management, transparent cost reporting, and milestone-based tracking, all of which Skydome Designs excels at providing.
How does Skydome Designs incorporate sustainability and green building principles into pharmaceutical facility design in Dubai?
Skydome Designs prioritizes sustainability by selecting environmentally friendly and durable materials with low VOCs and reduced embodied energy. We design for optimal energy efficiency through advanced energy-efficient MEP systems, smart lighting, and building envelope optimization. We also focus on waste reduction strategies during both construction and operation, and integrate water conservation measures. Our designs align with Dubai’s green building initiatives (e.g., Estidama, LEED principles) to minimize environmental impact, enhance indoor air quality, and significantly lower the facility’s lifecycle costs.
Why is BIM (Building Information Modeling) absolutely essential for pharmaceutical facility design in Dubai?
BIM (Building Information Modeling) is essential for pharmaceutical facility design in Dubai because it drastically improves collaboration among all project stakeholders, enabling precise clash detection and resolution early in the design phase to prevent costly construction errors. It provides advanced visualization capabilities for virtual walkthroughs, enhances project efficiency, and reduces design errors. Furthermore, BIM supports Dubai’s strong push for digital delivery in construction, facilitating efficient lifecycle management, predictive maintenance through digital twins, and streamlined facility operations post-handover.
What is the typical timeframe for designing and constructing a pharmaceutical facility interior in Dubai?
The typical timeframe for designing and constructing a pharmaceutical facility interior in Dubai can vary significantly based on the facility’s size, complexity, and specific requirements (e.g., sterile manufacturing vs. warehousing). Generally, a project can range from 18 to 36 months or more, covering initial consultation, conceptual design, detailed engineering, procurement, construction, and critical validation/commissioning phases. Factors like regulatory approvals, equipment lead times, and client decision-making also influence the schedule. Skydome Designs specializes in efficient project management to optimize these timelines, boasting a 97% on-time delivery rate.
What kind of cleanroom classifications are typically required for pharmaceutical facilities in Dubai?
Pharmaceutical facilities in Dubai typically require various cleanroom classifications depending on the specific manufacturing process (e.g., sterile vs. non-sterile, APIs). These classifications are usually defined by international standards such as ISO 14644 (e.g., ISO Class 5, ISO Class 7, ISO Class 8) and often correspond to EU GMP Grades (e.g., Grade A, B, C, D). The design ensures appropriate pressure differentials, air changes per hour (ACH), and HEPA/ULPA filtration to maintain these critical environmental conditions and prevent contamination.
Skydome Designs Pvt Ltd
- 📞 Contact: +91 7299072144
- ✉️ Email: info@skydomedesigns.com