The landscape of healthcare and pharmaceuticals is rapidly evolving, with digital transformation at its forefront. In a bustling economic hub like Raipur, the pharmaceutical sector is experiencing unprecedented growth, attracting significant new investments. This surge in development is not just about expanding capacity; it’s about building state-of-the-art facilities that are technologically advanced, highly efficient, and rigorously compliant with global standards. However, navigating the intricate complexities of designing and executing digital pharmaceutical facility interiors in Raipur presents a unique set of challenges. This comprehensive guide is crafted to provide you with expert, in-depth tips, helping you avoid common, costly mistakes and ensuring your pharmaceutical facility is not only future-proof but also fully compliant and optimized for success in 2025 and beyond.
The journey from conceptualization to a fully operational digital pharmaceutical facility is fraught with critical decisions, each carrying substantial implications for product quality, operational efficiency, regulatory adherence, and ultimately, your bottom line. An improperly designed interior can lead to bottlenecks, contamination risks, energy waste, and severe regulatory penalties. Conversely, a well-planned and executed design, particularly for Raipur’s digital pharmaceutical facility interiors, becomes a strategic asset, enabling faster time-to-market, enhanced productivity, and a robust framework for innovation.
The Indispensable Role of Expert Digital Pharmaceutical Facility Interiors in Raipur
With the fresh wave of investments pouring into Raipur, the imperative for robust and meticulously designed digital pharmaceutical facility interiors has never been greater. These aren’t just spaces; they are highly controlled ecosystems where precision, sterility, and efficiency converge. Expert design in this critical sector is fundamentally about de-risking your investment and accelerating project delivery. It’s about translating complex regulatory requirements and advanced technological needs into a functional, safe, and productive environment. Partnering with seasoned Raipur construction experts, specifically those with a deep understanding of pharmaceutical infrastructure, is not merely a recommendation; it is an absolute necessity to ensure your facility not only meets but often exceeds stringent industry standards and optimizes every facet of operational efficiency.
The consequences of poor design in pharmaceutical facilities are far-reaching and potentially catastrophic. They can range from significant regulatory non-compliance issues and the accompanying heavy fines and operational shutdowns, to compromised product quality, increased safety hazards for personnel, and a drastic decrease in overall productivity. In a digital pharmaceutical setting, where automation, data integrity, and interconnected systems are paramount, the interior design must seamlessly integrate these technologies without sacrificing functionality or compliance. This holistic approach ensures that the physical space supports the digital infrastructure, creating a synergy that drives innovation and maintains competitive advantage.
Why Choose Skydome Designs for Your Raipur Project?
When it comes to highly specialized projects like digital pharmaceutical facility interiors in Raipur, expertise, experience, and a proven track record are non-negotiable. Skydome Designs Pvt Ltd stands as a beacon of excellence in the architecture and interior design domain across India. While specializing in diverse sectors including hospital and healthcare interiors, residential, and retail projects, our particular prowess in designing and executing complex healthcare and pharmaceutical environments sets us apart. With nearly three decades of dedicated experience, we pride ourselves on delivering innovative, sustainable, and highly functional spaces that profoundly enhance user experiences and significantly boost operational efficiency. Our commitment to quality and client satisfaction is unwavering, making us the ideal partner for your ambitious Raipur project.
- 29+ years of unparalleled experience across India and various international locations, equipping us with a global perspective and local insight into pharmaceutical facility design.
- An impressive portfolio showcasing the successful delivery of 704+ digital pharmaceutical facility interiors assignments across Raipur, India, and globally over 24+ years, demonstrating our deep expertise and robust capacity.
- A fully integrated in-house team comprising highly skilled architects, specialized healthcare planners, experienced interior designers, and meticulous project managers. This multidisciplinary approach ensures seamless coordination and comprehensive solutions from concept to completion.
- Recipient of numerous award-winning designs, consistently delivering client-focused and sustainable solutions that adhere to the highest global standards. Our designs are not just aesthetically pleasing but are engineered for longevity, efficiency, and environmental responsibility.
- A formidable track record of projects consistently delivered on-time, on-budget, and conforming to the most stringent global standards, underpinned by an exceptional 99% on-time delivery rate, multi-disciplinary reviews, and dedicated post-occupancy support. This steadfast reliability is critical for complex pharmaceutical ventures in Raipur.
Embark on your journey to a world-class digital pharmaceutical facility in Raipur with Skydome Designs. Call us today for an expert consultation: +91 7299072144 | ✉️ Email: info@skydomedesigns.com
Key Mistakes to Avoid in Digital Pharmaceutical Facility Interior Design in Raipur
The planning and execution of digital pharmaceutical facility interiors in Raipur is an intricate process, where even minor oversights can lead to significant repercussions. To safeguard your investment and ensure long-term success, it is crucial to be aware of and proactively avoid these critical mistakes:
1. Ignoring Regulatory Compliance – The Cornerstone of Pharmaceutical Operations
Perhaps the most critical mistake to avoid is underestimating or failing to rigorously comply with the myriad of regulations governing pharmaceutical facilities. These facilities are subject to an exceptionally strict regulatory framework, both national and international, designed to ensure product safety, efficacy, and quality. Failing to incorporate these guidelines into the very fabric of your design can result in devastating consequences, including hefty fines, mandatory operational shutdowns, product recalls, and irreparable damage to your brand’s reputation. It is imperative that your design incorporates the latest guidelines from authoritative organizations such as the World Health Organization (WHO), Good Manufacturing Practices (GMP) standards (e.g., USFDA, EU GMP), and crucially, local Raipur and state-level authorities governing pharmaceutical manufacturing and healthcare infrastructure. A proactive approach involves engaging experts who are not only familiar with these regulations but also adept at anticipating future regulatory shifts, thereby future-proofing your facility in Raipur.
2. Overlooking the Critical Importance of MEP Services and Cleanroom Technology
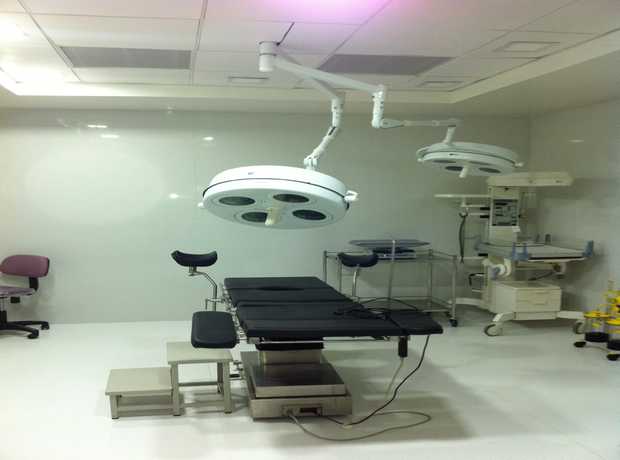
In a pharmaceutical facility, particularly a digital one, Mechanical, Electrical, and Plumbing (MEP) services are not merely utilities; they are the lifeblood that maintains the tightly controlled environments essential for manufacturing sterile and high-quality products. Insufficient, poorly designed, or outdated MEP systems can critically compromise product integrity, patient safety, and operational continuity. This includes the highly specialized Heating, Ventilation, and Air Conditioning (HVAC) systems crucial for maintaining precise temperature, humidity, and pressure differentials within cleanrooms. Advanced air filtration systems (HEPA filters), sophisticated building management systems (BMS), robust backup power solutions (generators, UPS), and meticulously designed process piping for utilities like purified water, WFI, and clean steam are non-negotiable. Furthermore, the integration of cutting-edge cleanroom technology, adhering to ISO classifications (e.g., ISO Class 5, 7, 8), requires expert knowledge in material selection, air change rates, unidirectional airflow, and personnel/material gowning procedures. Skydome Designs offers comprehensive, state-of-the-art MEP services and cleanroom design expertise, meticulously tailored to the specific and demanding needs of Raipur digital pharmaceutical facility interiors, ensuring a controlled, safe, and efficient operational environment.
3. Neglecting Workflow Optimization and Lean Principles
In any manufacturing environment, efficiency is directly linked to profitability, and in pharmaceutical production, it also impacts safety and compliance. An inefficient workflow can lead to bottlenecks, increased contamination risks due to unnecessary movement, higher operational costs, and reduced productivity. Designing your facility with a clear focus on minimizing movement, streamlining processes, and ensuring logical segregation of activities is paramount. This involves careful consideration of personnel flow (from entry to gowning, production, and exit), material flow (from raw materials to finished goods, minimizing cross-contamination), and waste flow. Employing lean manufacturing principles and utilizing advanced simulation tools during the design phase can help optimize layouts, reduce transit times, and prevent ergonomic issues. Skydome Designs excels in space planning expertise, meticulously analyzing operational requirements to optimize workflow and create highly efficient layouts for your Raipur digital pharmaceutical facility, ensuring smooth, logical, and compliant operations.
4. Disregarding Sustainable Construction Practices – A Vision for 2025
The global shift towards sustainability is not just an environmental imperative but a shrewd business strategy. Incorporating sustainable construction practices into your pharmaceutical facility design in Raipur offers a multitude of benefits that extend far beyond environmental responsibility. It leads to substantial long-term cost savings through reduced energy consumption (e.g., high-efficiency lighting, optimized HVAC systems, solar power integration), lower water usage (rainwater harvesting, water recycling systems), and minimized waste generation. Beyond the economic advantages, green building practices significantly enhance your company’s reputation, attracting environmentally conscious talent and investors, and demonstrating corporate social responsibility. Furthermore, sustainable design can contribute to a healthier and more comfortable work environment, leading to increased employee morale and productivity. Skydome Designs is a staunch advocate for environmental stewardship, prioritizing and integrating sustainable solutions into every aspect of digital pharmaceutical facility interiors in Raipur, helping clients achieve certifications like LEED and promoting a greener future for pharmaceutical manufacturing.
5. Lack of Proper Structural Engineering – Foundation for Safety and Future Growth
The structural integrity of a pharmaceutical facility is foundational to its safety, longevity, and operational stability. Failing to adequately address critical structural considerations can lead to severe operational disruptions, costly repairs, or, in extreme cases, catastrophic failures. Pharmaceutical facilities often house heavy machinery, vibration-sensitive equipment, and require specific load-bearing capacities for process floors and utility mezzanines. Consideration must also be given to local geological conditions, seismic activity (relevant for parts of India including Chhattisgarh), and the potential for future expansion or modification. Engaging a professional structural engineer with specialized experience in industrial and healthcare facilities, who deeply understands local building codes and environmental conditions in Raipur, is absolutely critical. This expertise ensures that the building’s framework can safely support all operational requirements, including the dynamic loads of equipment, controlled environment infrastructure, and future scalability. Skydome Designs integrates robust structural engineering expertise from the outset, ensuring a secure and resilient foundation for your pharmaceutical investment in Raipur.
6. Underestimating the “Digital” in Digital Pharmaceutical Facilities
The term “digital pharmaceutical facility” implies far more than just using computers. A significant mistake is failing to fully conceptualize and integrate advanced digital technologies into the very design and workflow of the facility. This includes neglecting infrastructure for the Internet of Things (IoT) sensors, real-time data analytics platforms, automation and robotics for manufacturing and material handling, Artificial Intelligence (AI) for quality control, and advanced Building Management Systems (BMS) that integrate environmental controls with process monitoring. The interior design must accommodate complex cabling, server rooms with appropriate cooling and security, flexible layouts for robotic arms, and ergonomic workstations for data-driven decision-making. Failing to plan for these digital layers from day one means a facility that is quickly outdated, inefficient, and unable to leverage the full potential of Industry 4.0. Skydome Designs specializes in designing environments that are truly “smart,” ensuring that the physical space empowers digital transformation within your Raipur pharmaceutical facility.
7. Inadequate Consideration of Cleanroom Material Selection and Finishes
Cleanroom integrity is paramount in pharmaceutical manufacturing. A common oversight is the selection of inappropriate materials and finishes for walls, floors, ceilings, and fixtures within controlled environments. These materials must be non-shedding, non-porous, easily cleanable, resistant to chemicals and sanitizers, and durable enough to withstand frequent cleaning cycles. Mistakes in material choice can lead to particle generation, microbial growth, and difficulty in maintaining required cleanliness classifications, directly impacting product quality and regulatory compliance. Consideration must be given to coving, seamless flooring (e.g., epoxy, vinyl), flush-mounted doors and windows, and specialized light fixtures that minimize dust accumulation and facilitate cleaning. Skydome Designs brings extensive experience in specifying and implementing ISO-compliant cleanroom materials and finishes, ensuring that every surface contributes to maintaining the highest standards of sterility in your digital pharmaceutical facility interiors in Raipur.
8. Neglecting Human Factors and Ergonomics
While technology and compliance are critical, the human element cannot be overlooked. A common mistake is designing facilities without adequate consideration for the well-being, comfort, and safety of the personnel who will work within them. Poor ergonomics can lead to physical strain, fatigue, errors, and reduced productivity. This includes inadequate lighting (both natural and artificial), poor acoustics, lack of comfortable break areas, and workstations that are not adjustable or designed for repetitive tasks. Designing with human factors in mind involves creating spaces that minimize physical stress, optimize visual comfort, provide sufficient space for movement, and incorporate biophilic design elements where appropriate. A happy, healthy, and comfortable workforce is more productive and less prone to mistakes. Skydome Designs understands that a successful facility integrates advanced technology with an environment that prioritizes employee well-being, enhancing both efficiency and job satisfaction in your Raipur pharmaceutical operations.
9. Insufficient Planning for Future Expansion and Adaptability
The pharmaceutical industry is dynamic, characterized by rapid technological advancements, evolving regulatory landscapes, and shifting market demands. A significant mistake is designing a facility that lacks the flexibility or scalability to adapt to these changes. Building a static facility that cannot accommodate new processes, additional equipment, or increased production volumes without extensive, costly renovations will quickly become a liability. Future-proofing involves modular design principles, accessible utility corridors, provisions for additional cleanroom capacity, and layouts that can be easily reconfigured. Planning for adaptability from the outset can save significant capital expenditure and downtime in the long run. Skydome Designs incorporates strategic planning for future growth and technological integration, ensuring your digital pharmaceutical facility in Raipur remains relevant and efficient for decades to come.
10. Overlooking Biosafety and Containment Measures
For facilities dealing with specific types of pharmaceutical production, such as biologics, vaccines, or active pharmaceutical ingredients (APIs), adequate biosafety and containment measures are absolutely non-negotiable. A critical mistake is failing to design appropriate biosafety levels (BSL-1 to BSL-4) into the facility’s interior, including specialized air handling, entry/exit protocols, decontamination showers, and waste management systems. The design must ensure the containment of hazardous biological agents or potent compounds to protect both personnel and the environment. This involves careful zoning, pressure cascade systems, and robust material specifications that can withstand rigorous decontamination procedures. Skydome Designs possesses the specialized knowledge required to integrate comprehensive biosafety and containment strategies into Raipur digital pharmaceutical facility interiors, adhering to the highest international standards for safe and secure operations.
How Skydome Designs Can Help with Your Raipur Pharmaceutical Facility
Choosing the right partner for your digital pharmaceutical facility interiors in Raipur is the most critical decision you will make. Skydome Designs offers an unparalleled suite of services, backed by decades of specialized experience, to ensure your project is a resounding success. We don’t just design spaces; we engineer environments that are compliant, efficient, sustainable, and future-ready.
- Comprehensive Design-Build Solutions: We offer integrated design-build solutions that provide a seamless, single-point responsibility approach for project execution. This minimizes coordination complexities, accelerates timelines, and ensures cost predictability, which is crucial for intricate pharmaceutical projects.
- BIM-Led Coordination and Value Engineering: Our projects benefit from advanced BIM-led coordination, enabling precise planning, clash detection, and optimized material usage, significantly reducing errors and waste. This is complemented by strategic value engineering to maximize cost-effectiveness without compromising quality or compliance. Every aspect of design and construction is subjected to rigorous quality control, meticulously tailored to the specific regulatory and environmental context of Raipur.
- Adherence to Global Design Standards with Local Code Expertise: Skydome Designs prides itself on developing designs that meet the most stringent global pharmaceutical design standards (e.g., WHO GMP, USFDA, EU GMP) while also possessing intimate knowledge of local building codes, regulations, and environmental factors specific to Raipur. This dual expertise ensures both international competitiveness and local compliance.
- Expertise in Creating Controlled Environments and Cleanrooms: Our team has extensive, proven experience in designing and implementing highly specialized controlled environments and cleanrooms, critical for pharmaceutical manufacturing. This includes meticulous planning for air filtration, pressure differentials, material selection, and sophisticated HVAC systems to meet various ISO classifications and maintain aseptic conditions.
- Strategic Project Management and Timely Delivery: With a track record of 99% on-time delivery across 704+ digital pharmaceutical facility interiors assignments globally, our project management methodologies are robust and highly effective. We emphasize multi-disciplinary reviews and provide dedicated post-occupancy support, ensuring that the outcomes not only meet but exceed expectations, underpinning the long-term success of your facility in Raipur.
Ready to transform your vision into a compliant and cutting-edge pharmaceutical reality? Contact Skydome Designs today to discuss your project and leverage our extensive experience: +91 7299072144 | ✉️ Email: info@skydomedesigns.com
Our Expertise in Raipur and Beyond
Our expansive portfolio demonstrates the successful delivery of 704+ digital pharmaceutical facility interiors assignments across Raipur, India, and globally over 24+ years. This vast experience ensures we bring a depth of knowledge to every project. Our commitment to on‑time delivery, achieving an impressive 99% success rate, is a testament to our meticulous planning, multi‑disciplinary reviews, and unwavering post‑occupancy support, all of which underpin successful outcomes. We possess an intimate understanding of the nuances of local regulations in Raipur, combined with a global perspective on best practices. This ensures that your facility is designed not only to meet present requirements but also to anticipate future challenges and opportunities. Our dedicated team understands the critical importance of timely project completion in the fast-paced pharmaceutical industry, minimizing downtime and maximizing your return on investment.
Don’t compromise on expertise. Get your digital pharmaceutical facility project in Raipur started with a proven leader. Contact Skydome Designs today for a consultation tailored to your unique needs! +91 7299072144 | ✉️ Email: info@skydomedesigns.com
Deep Dive: The “Digital” Aspect in Pharmaceutical Facility Interiors
The transition to a “digital” pharmaceutical facility is not merely an upgrade; it’s a paradigm shift. In 2025, a truly digital facility in Raipur will embody the principles of Industry 4.0, integrating advanced technologies at every level to enhance efficiency, data integrity, and regulatory compliance. The interior design must be conceived to support and amplify these digital capabilities:
Integrated Automation and Robotics
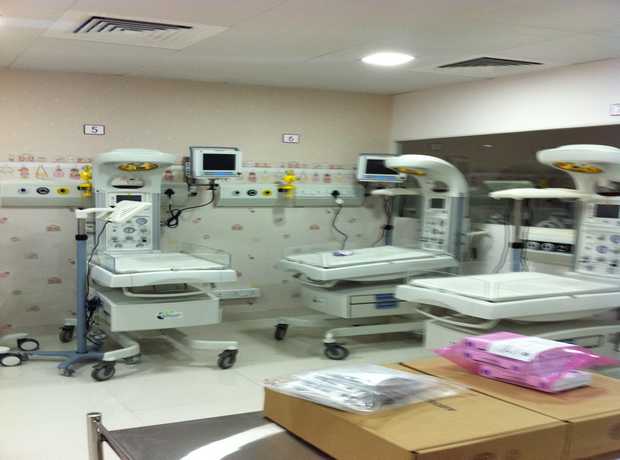
Modern pharmaceutical facilities are increasingly reliant on automation for tasks ranging from raw material handling to precise dosing, packaging, and quality control. The interior must provide appropriate spatial layouts for robotic arms, automated guided vehicles (AGVs), and conveyor systems, ensuring smooth, unobstructed pathways and zones for automated processes. Flooring must be robust enough for heavy loads and continuous movement, and electrical/data infrastructure must be omnipresent to power and communicate with these intelligent machines. Cleanroom environments must seamlessly integrate automated systems without compromising sterility, requiring careful consideration of robotic arm materials, air movement patterns, and access points for maintenance.
IoT and Real-time Monitoring
The “digital” aspect hinges on data. Interior design must facilitate the strategic placement of IoT sensors for continuous, real-time monitoring of critical parameters such as temperature, humidity, pressure, particulate matter, and microbial counts. These sensors require unobtrusive integration into walls, ceilings, and equipment, with robust data cabling or wireless networks. The design should also incorporate control rooms or monitoring stations where this influx of data can be visualized and analyzed, enabling immediate adjustments and proactive maintenance. For Raipur digital pharmaceutical facility interiors, this means an infrastructure that supports predictive analytics and smart alerts, enhancing operational transparency and regulatory reporting.
Advanced Data Management and Cybersecurity Infrastructure
A digital facility generates colossal amounts of data, from manufacturing parameters to quality control results and environmental monitoring. The interior design must allocate secure, climate-controlled spaces for servers, data storage units, and network infrastructure. These areas require specialized HVAC, fire suppression systems, and access controls to protect sensitive data. Furthermore, the entire facility’s network architecture, from wiring to wireless access points, must be designed with cybersecurity as a primary concern, safeguarding against breaches and ensuring data integrity, which is vital for GMP compliance. Skydome Designs ensures that the physical infrastructure supports a secure and robust digital backbone for your Raipur operations.
Virtual Reality (VR) and Augmented Reality (AR) for Training and Maintenance
Looking ahead to 2025, digital pharmaceutical facilities will increasingly leverage VR and AR for immersive training simulations, complex maintenance procedures, and remote troubleshooting. The interior design should consider how these technologies will be utilized. This might include dedicated spaces for VR training, or simply ensuring ample, well-lit spaces where AR overlays can be effectively used during operational tasks, providing technicians with real-time diagnostic information on equipment. This digital integration enhances safety, reduces human error, and improves efficiency.
Human-Machine Interface (HMI) and Ergonomic Digital Workstations
Even with automation, human interaction with digital systems remains crucial. The interior design must incorporate ergonomic workstations equipped with high-resolution monitors, intuitive HMIs, and comfortable seating, reducing operator fatigue during long shifts. These digital control points should be strategically located to provide optimal visibility and access to production areas while maintaining cleanroom protocols. The lighting design around these workstations is also critical to minimize glare and eye strain, promoting accuracy and efficiency in data entry and monitoring.
Essential Considerations for Cleanroom Design in Raipur’s Digital Pharma Facilities
Cleanrooms are the heart of any pharmaceutical facility, especially those involved in sterile or aseptic manufacturing. Their design, construction, and operation are subject to extremely rigorous standards to prevent contamination. For digital pharmaceutical facility interiors in Raipur, the cleanroom is where precision engineering meets environmental control:
ISO Classification and Air Filtration Systems
Cleanrooms are classified based on the number of particles allowed per cubic meter of air (e.g., ISO Class 5, 7, 8). Achieving and maintaining these classifications requires sophisticated HVAC systems equipped with multi-stage filtration, including HEPA (High-Efficiency Particulate Air) or ULPA (Ultra-Low Penetration Air) filters. The interior design must accommodate the substantial space required for these air handling units, ducting, and filter banks. Airflow patterns, often unidirectional or laminar, are critical and dictate ceiling plenum design, return air grilles, and equipment placement to sweep particles away from critical zones.
Pressure Differentials and Zoning
Maintaining positive or negative pressure differentials between cleanroom zones is crucial to control airflow and prevent contamination ingress or egress. The interior design must clearly delineate these zones – from “black” (non-classified) to “grey” (controlled non-classified) to “white” (classified cleanroom) areas – and incorporate airlocks and interlocked doors to manage pressure cascades effectively. The physical layout directly influences the effectiveness of these pressure regimes, requiring careful space planning and consideration of personnel and material flows.
Material Selection and Finishes for Aseptic Environments
Every surface within a cleanroom must be carefully selected to prevent particle shedding, microbial growth, and facilitate rigorous cleaning and sanitization. This includes:
- Walls: Non-shedding, smooth, seamless, and chemical-resistant materials like epoxy-coated panels, PVC-laminated panels, or stainless steel. Coving at wall-to-floor and wall-to-ceiling junctions is essential to eliminate sharp corners where dust and microbes can accumulate.
- Flooring: Seamless, impervious, anti-static, and chemical-resistant options such as epoxy resin, vinyl, or specialized rubber. It must be able to withstand heavy traffic and frequent cleaning.
- Ceilings: Often walk-on or accessible plenum ceilings, constructed from non-shedding, washable panels with integrated, flush-mounted light fixtures and utility ports to prevent particle traps.
- Doors and Windows: Flush-mounted, airtight, and often interlocked to maintain pressure differentials. Double-glazed windows with flush frames are typical.
Skydome Designs’ expertise ensures that all material specifications for digital pharmaceutical facility interiors in Raipur meet the highest cleanroom standards, balancing durability, compliance, and aesthetic appeal.
Utilities and Service Integration
Cleanrooms require a complex network of utilities, including purified water (PW), water for injection (WFI), clean steam, process gases (e.g., nitrogen, compressed air), and specialized electrical outlets. The interior design must integrate these services efficiently, often through utility chases, service panels, or overhead grids, ensuring accessibility for maintenance without compromising the cleanroom envelope. The design must also account for waste disposal systems that comply with cleanroom protocols and environmental regulations.
Future-Proofing Your Digital Pharmaceutical Facility in Raipur for 2025 and Beyond
Building a pharmaceutical facility is a significant long-term investment. To ensure its continued relevance and profitability, it must be future-proofed against technological advancements, regulatory changes, and market shifts. For Raipur digital pharmaceutical facility interiors, this means designing with foresight:
Modularity and Flexibility
The core design principle for future-proofing is modularity. Spaces should be designed in adaptable modules that can be easily reconfigured or expanded without disrupting ongoing operations. This involves using non-load-bearing partitions where possible, standardized utility connections, and flexible layouts for equipment placement. As new manufacturing technologies emerge (e.g., continuous manufacturing, personalized medicine), a modular design allows for seamless integration and adaptation.
Scalable Infrastructure
Anticipate future growth. Design the facility’s infrastructure – power, HVAC capacity, water purification systems, and data networks – with scalability in mind. Oversizing certain components or providing space for future additions (e.g., empty conduits, spare panel capacity) can significantly reduce the cost and complexity of future upgrades. This forethought is critical for dynamic industries like pharmaceuticals in Raipur.
Digital Adaptability
Ensure the facility’s digital infrastructure can evolve. This means using open-architecture control systems, standard communication protocols, and physical spaces that can accommodate new sensors, robotics, and data processing equipment. A truly future-proof digital facility embraces continuous technological integration rather than being confined to current capabilities. Skydome Designs builds in this digital adaptability from the conceptual stage.
Regulatory Foresight
Regulations are constantly evolving. An expert design firm like Skydome Designs stays abreast of upcoming regulatory changes and designs facilities that can easily adapt to new compliance requirements. This might involve additional segregation needs, enhanced monitoring capabilities, or new documentation standards. Building in this foresight avoids costly retrofits and ensures continuous compliance for your Raipur pharmaceutical facility.
Sustainability for Long-Term Value
Sustainable design is inherently future-proof. Facilities designed with energy efficiency, water conservation, and waste reduction in mind will be more resilient to rising utility costs and increasingly stringent environmental regulations. Furthermore, sustainable operations enhance corporate image and attract top talent, securing long-term value for your investment in Raipur.
FAQ: Digital Pharmaceutical Facility Interiors in Raipur
Here are some frequently asked questions about designing and constructing digital pharmaceutical facility interiors in Raipur, addressing common concerns and providing expert insights:
What are the absolute key considerations for designing a digital pharmaceutical facility in Raipur for 2025?
The absolute key considerations are multi-faceted: paramount is stringent regulatory compliance (local, national, and international GMPs), meticulously designed MEP services for environmental control and critical utilities, optimized workflow pathways for personnel and materials, the integration of advanced digital technologies (automation, IoT, data management), robust structural integrity for safety and longevity, and a strong commitment to sustainable construction practices. Partnering with an experienced Raipur digital pharmaceutical facility interiors company like Skydome Designs, which offers BIM-led coordination and global design standards with local code expertise, is crucial to navigate these complexities successfully.
How can I ensure my pharmaceutical facility in Raipur meets all necessary regulatory requirements, including GMP standards?
To ensure full regulatory compliance, you must engage a design and construction firm with a deep and proven understanding of both local Raipur regulations and international standards such as WHO GMP, USFDA, and EU GMP. They should have an extensive track record in navigating complex approval processes and a rigorous quality control system. Skydome Designs, with its 29+ years of experience and 704+ assignments delivered, possesses this specific expertise, ensuring that every design aspect of your Raipur digital pharmaceutical facility interiors adheres to the strictest compliance mandates from the very first sketch.
What are the most significant benefits of investing in sustainable construction for pharmaceutical facilities in Raipur?
Investing in sustainable construction for your pharmaceutical facility in Raipur yields numerous significant benefits. These include substantial long-term operational cost reductions through enhanced energy efficiency (lighting, HVAC), optimized water usage, and reduced waste disposal fees. Environmentally, it minimizes your facility’s ecological footprint. From a business perspective, it greatly enhances your company’s reputation, attracting top talent and socially conscious investors, while also providing a healthier, more productive work environment for your employees. Skydome Designs integrates these sustainable solutions, ensuring your Raipur pharmaceutical facility is future-proof and economically advantageous.
How does Skydome Designs integrate advanced digital technologies into its pharmaceutical facility interior designs in Raipur?
Skydome Designs excels in integrating advanced digital technologies by designing the core infrastructure to support Industry 4.0. This includes meticulous planning for seamless automation and robotics pathways, robust IoT sensor placement for real-time monitoring, secure data centers for advanced analytics, and ergonomic workstations for human-machine interfaces. We ensure that the physical layout of your digital pharmaceutical facility interiors in Raipur is optimized to accommodate high-speed data networks, power supply for cutting-edge equipment, and flexible spaces that can adapt to future technological advancements, ensuring your facility is truly smart and efficient.
How much does it typically cost to design and build a state-of-the-art digital pharmaceutical facility in Raipur, and what factors influence this cost?
The cost to design and build a state-of-the-art digital pharmaceutical facility in Raipur can vary significantly, typically ranging from several crores to hundreds of crores of Indian Rupees, depending on numerous factors. Key cost drivers include the overall size and complexity of the facility, the required ISO cleanroom classifications, the level of automation and digital integration desired, the choice of materials (especially for cleanrooms), the extent of specialized MEP services (e.g., high-purity water systems, complex HVAC), site preparation requirements, and specific regulatory compliance measures. For a detailed and accurate cost estimate tailored to your specific project needs and objectives, we highly recommend contacting Skydome Designs for a comprehensive consultation. Our value engineering approach ensures optimal investment without compromising quality.
What is BIM-led coordination, and why is it important for pharmaceutical facility design in Raipur?
BIM-led coordination, or Building Information Modeling-led coordination, is a highly advanced process where a digital 3D model of the entire facility is created, integrating architectural, structural, and MEP designs. This holistic model allows for early clash detection, design optimization, and precise visualization before physical construction begins. For pharmaceutical facility design in Raipur, BIM-led coordination is critically important because it minimizes errors, reduces costly rework, enhances collaboration among all stakeholders, ensures precise material quantities, and ultimately accelerates project timelines and maintains budgets. Skydome Designs leverages this technology extensively, alongside meticulous quality control tailored to Raipur, to deliver superior outcomes for your complex projects.
Can Skydome Designs provide design-build solutions for digital pharmaceutical facilities in Raipur?
Yes, Skydome Designs specializes in providing comprehensive design-build solutions for digital pharmaceutical facility interiors in Raipur. This integrated approach means we manage the entire project lifecycle, from initial conceptual design and regulatory approvals to detailed engineering, construction, and commissioning. This single-point responsibility streamlines communication, reduces administrative burdens, ensures greater cost predictability, and significantly accelerates project delivery. Our design-build model, backed by a 99% on-time delivery rate, is ideal for complex pharmaceutical projects requiring stringent quality control and seamless execution.
Conclusion
The endeavor of designing and building a state-of-the-art digital pharmaceutical facility in Raipur is a multifaceted undertaking that demands meticulous planning, profound expertise, and flawless execution. As Raipur’s pharmaceutical sector continues its dynamic expansion towards 2025 and beyond, the critical distinction between success and costly setbacks often hinges on the quality of your interior design and construction partner. By proactively identifying and avoiding the common pitfalls discussed in this guide – from neglecting stringent regulatory compliance and critical MEP services to underestimating the “digital” aspect and failing to plan for future adaptability – you can lay a robust foundation for your investment.
Partnering with experienced, specialized professionals like Skydome Designs is not merely an option; it is a strategic imperative. Our nearly three decades of dedicated experience, evidenced by the successful delivery of 704+ digital pharmaceutical facility interiors assignments across diverse geographies including Raipur, underscore our unparalleled capability. Our commitment to BIM-led coordination, value engineering, and rigorous quality control, coupled with an exceptional 99% on-time project delivery rate, ensures that your facility will be compliant, exceptionally efficient, technologically advanced, and inherently future-proof. Don’t risk costly errors, delays, or non-compliance; invest wisely in expert design and execution services from the very inception of your project.
Ready to elevate your pharmaceutical manufacturing capabilities in Raipur? Contact Skydome Designs, your trusted and experienced Raipur digital pharmaceutical facility interiors company, today to begin your journey towards a world-class facility! Call us now: +91 7299072144 | ✉️ Email: info@skydomedesigns.com
About Skydome Designs
Skydome Designs Pvt Ltd is a premier architecture and interior design firm based in India, renowned for its specialization in highly complex hospital and healthcare interiors, alongside distinctive residential and retail projects. With nearly 30 years of extensive expertise, our firm is committed to delivering innovative, sustainable, and highly functional spaces. We pride ourselves on crafting environments that not only enhance experiences but also significantly boost operational efficiency, consistently exceeding client expectations and setting new industry benchmarks. Our multidisciplinary team ensures every project benefits from a holistic and expertly managed approach.
Learn more about Skydome Designs and explore our extensive portfolio of successful projects.