The pharmaceutical industry in New York stands at a pivotal juncture. With innovation accelerating, regulatory scrutiny intensifying, and a global demand for quality rising, ensuring your pharmaceutical facility in New York meets and exceeds all current and future requirements is not just good practice – it’s an imperative for sustained success and market leadership. This comprehensive blog post delves deep into essential checklists for pharmaceutical facility interiors in New York for 2025, meticulously focusing on compliance, unwavering quality, future-ready design, and operational excellence.
The landscape of pharmaceutical manufacturing and research is evolving rapidly. New York, a hub of biotech and pharmaceutical innovation, is leading the charge in embracing advanced technologies, sustainable practices, and patient-centric designs. As we approach 2025, facility owners and developers must strategically integrate these elements to not only satisfy immediate regulatory demands but also to secure long-term operational efficiency and competitive advantage. Our detailed checklist will guide you through crucial considerations, helping you lower lifecycle costs, optimize performance, and create a truly cutting-edge pharmaceutical facility in the Empire State.
Understanding the Dynamic Landscape of Pharmaceutical Facility Interiors in New York
New York’s pharmaceutical sector is a complex ecosystem, characterized by its dynamism, innovation, and stringent oversight. The state’s strategic location, access to world-class research institutions, and a highly skilled workforce make it an attractive location for pharmaceutical companies. However, this competitive environment also necessitates a meticulous approach to facility design and construction. The focus isn’t merely on erecting structures, but on crafting highly controlled environments that are conducive to the precise, sterile, and compliant production of life-saving medications.
Staying ahead of the curve means understanding not just today’s regulations but anticipating tomorrow’s. This includes a forward-looking perspective on evolving FDA guidelines, specific New York State Department of Health (NYSDOH) mandates, local building codes, and international quality standards. Beyond regulation, market trends such as personalized medicine, biologics, and advanced therapy medicinal products (ATMPs) are influencing facility design, demanding greater flexibility, modularity, and rapid adaptation capabilities. Furthermore, the state’s increasing emphasis on environmental stewardship and technological integration, particularly in sustainability and digital delivery, requires innovative design and construction approaches that transcend conventional methodologies. A holistic view, integrating architectural design, engineering, compliance, and operational workflow, is essential for a pharmaceutical facility interior that truly excels in New York.
The Growing Importance of Specialized Expertise in New York
Navigating this intricate landscape requires more than just general construction knowledge; it demands specialized expertise in pharmaceutical facility design. This encompasses an understanding of sterile processing, containment strategies, advanced HVAC systems, critical utility infrastructure, and material science specific to pharmaceutical applications. Engaging a partner with a deep portfolio in New York’s pharmaceutical industry ensures that your project benefits from proven strategies, local insights, and a track record of successful regulatory navigation. Such a partner can translate complex requirements into practical, cost-effective, and compliant interior solutions.
Comprehensive Checklists for Pharmaceutical Facility Interiors in New York (2025)
Success in pharmaceutical facility design hinges on a multi-faceted approach. We’ve broken down the critical elements into comprehensive checklists to ensure every aspect of your New York facility’s interior is meticulously planned and executed.
1. Regulatory Compliance Checklist: The Foundation of Trust and Operation
Adhering to regulatory mandates is the bedrock of any pharmaceutical operation. Non-compliance can lead to severe penalties, operational shutdowns, and irreparable damage to reputation. This checklist covers critical aspects of compliance, ensuring your facility is not just operational, but also legally sound and ethically responsible:
- FDA Guidelines: Current Good Manufacturing Practices (cGMP):
- Ensure all aspects of facility design and construction comply with FDA’s cGMP regulations (21 CFR Parts 210 and 211). This includes detailed provisions for facility layout, environmental controls, material flow, personnel flow, segregation of operations, and prevention of cross-contamination.
- Verify that specific areas for raw material handling, manufacturing, packaging, warehousing, and quality control are clearly defined and appropriately designed to prevent mix-ups and errors.
- Implement design features that facilitate cleaning, maintenance, and pest control, minimizing areas where dust or contaminants can accumulate.
- Document all design decisions, material selections, and construction processes to demonstrate cGMP adherence during audits.
Skydome Designs possesses extensive knowledge of FDA cGMP regulations and leverages over two decades of experience to help clients navigate the complex regulatory landscape in New York, ensuring every design detail aligns with compliance requirements.
- New York State Regulations: Specific Mandates for Pharmaceutical Facilities:
- Familiarize yourself with specific regulations imposed by the New York State Department of Health (NYSDOH) and other relevant state agencies concerning pharmaceutical manufacturing, storage, and distribution.
- Comply with local building codes, zoning ordinances, fire safety regulations (e.g., from the New York State Department of State, Division of Code Enforcement and Administration), and electrical codes specific to the municipalities in New York where your facility is located.
- Address specific requirements related to controlled substances if applicable, collaborating with the New York State Bureau of Narcotic Enforcement.
- Ensure compliance with all permits and licensing requirements from local and state authorities before construction begins and throughout operation.
- Environmental Regulations: Protecting the Planet and Your Operation:
- Comply with federal Environmental Protection Agency (EPA) regulations and specific New York State Department of Environmental Conservation (NYSDEC) requirements regarding hazardous waste disposal, air emissions, wastewater treatment, and storm water management.
- Design interior spaces to facilitate safe and compliant handling and storage of chemicals and hazardous materials, including appropriate ventilation, spill containment, and emergency response infrastructure.
- Integrate systems for monitoring and reporting environmental impact, aligning with New York’s commitment to environmental protection.
- Consider obtaining relevant environmental certifications, demonstrating your commitment to ecological responsibility.
- Safety Standards: Safeguarding Personnel and Operations:
- Adhere to stringent safety standards outlined by the Occupational Safety and Health Administration (OSHA) and New York State Public Employee Safety and Health Bureau (PESH) to protect personnel from workplace hazards.
- Design for adequate egress paths, emergency showers, eye wash stations, and fire suppression systems (sprinklers, alarms) that meet or exceed NFPA (National Fire Protection Association) standards.
- Incorporate ergonomic design principles into workspaces to prevent repetitive strain injuries and enhance employee comfort and productivity.
- Implement robust security measures, including access control, surveillance systems, and secure storage for sensitive materials, to protect assets and intellectual property.
Achieving a 100% compliant facility in New York requires a deep understanding of multi-layered regulations. Skydome Designs offers comprehensive regulatory compliance consulting as part of its end-to-end delivery service, ensuring your pharmaceutical interior project meets all federal, state, and local standards.
2. Quality Control Checklist: The Heart of Pharmaceutical Excellence
The primary objective of a pharmaceutical facility is to consistently produce high-quality, safe, and effective products. The interior design plays a crucial role in preventing contamination, ensuring product integrity, and facilitating rigorous quality control. This checklist focuses on critical quality control measures:
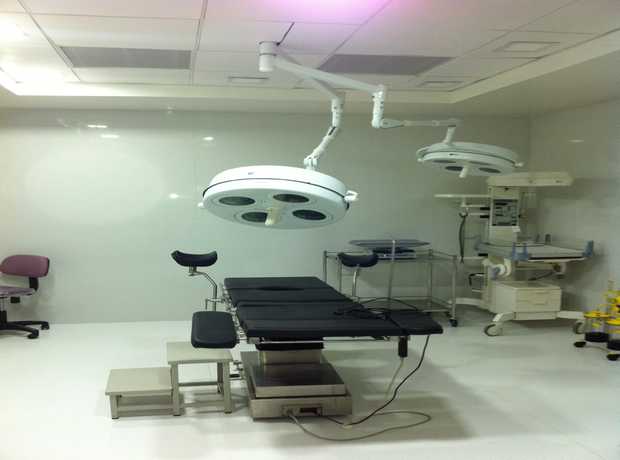
- Cleanroom Design and Construction: Precision for Purity:
- Ensure cleanrooms meet the required ISO classifications (e.g., ISO 5, 7, 8) based on the manufacturing process and product sensitivity, with meticulously planned air filtration systems (HEPA/ULPA filters).
- Implement precise pressure differentials and airlock systems to control airflow direction, preventing the ingress of contaminants between different cleanroom grades and to less-controlled areas.
- Design smooth, non-porous, and easily cleanable surfaces for walls, floors, and ceilings to minimize particle generation and microbial accumulation.
- Incorporate gowning and de-gowning rooms that are appropriately configured to maintain cleanroom integrity and facilitate aseptic practices.
Consider professional cleanroom design services that understand the nuances of ISO classification and contamination control for optimal quality control and compliance. (Outbound Authority Link)
- Material Selection: The Unsung Hero of Cleanliness:
- Choose building materials that are non-reactive, non-shedding, chemical-resistant (to cleaning agents and product ingredients), and resistant to microbial growth.
- Prioritize materials with low particulate generation, smooth finishes, and seamless installation to avoid crevices and difficult-to-clean areas. Examples include epoxy floors, coated panels, and stainless steel fixtures.
- Verify material compatibility with the specific manufacturing processes, temperature ranges, and humidity levels anticipated within the facility.
- Document all material specifications and certifications for traceability and regulatory submission.
- HVAC Systems: The Air You Breathe, The Quality You Ensure:
- Implement robust Heating, Ventilation, and Air Conditioning (HVAC) systems designed specifically for pharmaceutical environments, capable of precise control over temperature, humidity, and air quality.
- Ensure HVAC systems incorporate multi-stage filtration, including pre-filters, medium-efficiency filters, and terminal HEPA/ULPA filters, to achieve required particulate cleanliness levels.
- Design for appropriate air changes per hour (ACH) specific to each classified area, ensuring efficient removal of airborne contaminants.
- Integrate sophisticated building management systems (BMS) to monitor and control HVAC parameters in real-time, with alarms for out-of-spec conditions and data logging for audit trails.
- Equipment Validation: Proving Performance and Purity:
- Validate all critical manufacturing, laboratory, and utility equipment (e.g., sterilization equipment, purified water systems, HVAC components) to ensure it performs as intended and consistently meets quality standards (Installation Qualification, Operational Qualification, Performance Qualification – IQ, OQ, PQ).
- Design interior layouts that facilitate easy access for equipment maintenance, calibration, and cleaning, minimizing downtime and ensuring operational continuity.
- Ensure utility supply points (e.g., WFI, clean steam, compressed air, gases) are strategically located and validated to deliver consistent quality and quantity to process equipment.
- Provide adequate space for equipment staging, maintenance, and future expansion without compromising operational flow or cleanroom integrity.
A commitment to impeccable quality is a hallmark of industry leaders. Let Skydome Designs apply its 24+ years of expertise in delivering pharmaceutical facility interiors with meticulous quality control, setting new benchmarks for your New York operations. Our multi‑disciplinary reviews ensure every detail is scrutinized.
3. Sustainability Checklist: Building a Greener, More Efficient Future
Sustainability is no longer a niche concern; it’s a core component of responsible business and a driver of long-term cost savings. New York State is a leader in promoting green initiatives, and integrating sustainable practices into pharmaceutical facility design can significantly reduce environmental impact and operational costs. This checklist covers key sustainability considerations for 2025 and beyond:
- Energy Efficiency: Cutting Costs, Reducing Carbon:
- Implement energy-efficient lighting systems, such as LED technology, with occupancy sensors and daylight harvesting controls to reduce electricity consumption.
- Optimize HVAC system design for energy efficiency, utilizing variable frequency drives (VFDs), heat recovery systems, and advanced controls to minimize energy waste.
- Utilize high-performance building envelopes, including superior insulation, high-efficiency windows, and airtight construction, to reduce heating and cooling loads.
- Explore renewable energy sources, such as rooftop solar panels or integration with off-site renewable energy projects, to offset grid energy consumption.
- Water Conservation: Precious Resource Management:
- Utilize water-saving technologies and practices, including low-flow fixtures in restrooms and break areas, and water-efficient landscaping.
- Implement advanced water treatment and recycling systems for non-critical processes, such as cooling tower blowdown or utility water, to reduce overall water demand.
- Monitor water usage closely with smart metering systems to identify areas for further optimization and leakage detection.
- Design for efficient production of purified water (e.g., WFI) to minimize waste in a critical utility.
- Sustainable Materials: Eco-Conscious Construction:
- Choose building materials with recycled content, low volatile organic compound (VOC) emissions, and those sourced regionally to reduce transportation impact.
- Prioritize rapidly renewable materials and those with environmental product declarations (EPDs) or health product declarations (HPDs) that detail their environmental and health impacts.
- Consider the lifecycle impact of materials, focusing on durability, ease of maintenance, and recyclability at the end of the facility’s life.
- Utilize non-toxic, anti-microbial paints and finishes that contribute to better indoor air quality without compromising sterility.
- Waste Reduction: A Circular Economy Approach:
- Implement comprehensive waste reduction and recycling programs for both construction waste and operational waste, including hazardous and non-hazardous materials.
- Design dedicated spaces for waste segregation, compaction, and temporary storage to facilitate efficient disposal and recycling.
- Explore strategies for minimizing process waste through optimized manufacturing workflows and material utilization.
- Engage in waste-to-energy initiatives where feasible and compliant with New York State regulations.
Skydome Designs specializes in sustainable construction solutions for pharmaceutical facilities, integrating eco-friendly practices with regulatory compliance. Contact us to discuss your project and how we can help you achieve a greener, more cost-effective facility: +91 7299072144 or info@skydomedesigns.com.
4. Digital Delivery & BIM Checklist: The Future of Project Management
The digitalization of construction and facility management is transforming how pharmaceutical projects are conceived, delivered, and operated. Leveraging digital tools enhances efficiency, accuracy, and collaboration, leading to significant lifecycle cost reductions and improved asset performance. This checklist focuses on digital delivery and Building Information Modeling (BIM):
- BIM Implementation: The Digital Blueprint for Success:
- Utilize BIM throughout the entire project lifecycle, from conceptual design and detailed engineering to construction sequencing, facility management (FM), and potential de-commissioning.
- Develop comprehensive 3D models that integrate architectural, structural, MEP (Mechanical, Electrical, Plumbing), and process equipment information for clash detection and spatial coordination, minimizing costly on-site reworks.
- Leverage BIM for accurate quantity take-offs, cost estimation, and scheduling, improving budget control and project predictability.
- Employ BIM models for detailed visualization and stakeholder engagement, allowing for early feedback and design optimization.
Explore the benefits of BIM for pharmaceutical facilities, a cornerstone of efficient project delivery and long-term asset management. (Outbound Authority Link)
- Virtual Reality (VR) & Augmented Reality (AR): Immersive Design & Training:
- Employ VR for immersive design visualization, allowing clients and stakeholders to “walk through” the proposed facility interior before construction begins, identifying potential issues and refining layouts.
- Utilize AR on-site for overlaying BIM models onto physical construction, verifying progress, identifying discrepancies, and assisting with complex installations.
- Develop VR/AR applications for personnel training, simulating complex procedures, equipment operation, and emergency scenarios in a safe, virtual environment, enhancing preparedness and reducing risks.
- Data Analytics & IoT Integration: Smart Facilities, Optimized Performance:
- Implement Internet of Things (IoT) sensors throughout the facility to collect real-time data on environmental conditions (temperature, humidity, air pressure), equipment performance, energy consumption, and personnel movement.
- Use data analytics to optimize facility performance, predict maintenance needs (predictive maintenance), identify inefficiencies, and continuously improve operational workflows.
- Integrate data from various systems (BMS, LIMS, QMS) into a unified platform for comprehensive monitoring and informed decision-making.
- Leverage AI and machine learning algorithms to identify patterns, automate routine tasks, and provide proactive insights for facility management.
- Cloud-Based Collaboration: Seamless Teamwork, Anywhere:
- Utilize cloud-based platforms for seamless collaboration among project stakeholders, including owners, architects, engineers, contractors, and vendors, regardless of their geographical location.
- Ensure all project documentation, drawings, models, and communications are stored in a centralized, secure cloud environment, providing real-time access to the latest information.
- Implement robust data security protocols and access controls to protect sensitive project information and intellectual property.
- Facilitate digital handover packages, ensuring all critical facility information is readily available for operations and maintenance teams post-construction.
Skydome Designs is at the forefront of digital delivery, leveraging BIM, VR, and data analytics to provide end‑to‑end pharmaceutical facility interior solutions in New York. Experience transparent costs and milestone‑based reporting with our award‑winning team.
5. Human-Centric Design and Ergonomics: Enhancing Productivity and Well-being
Beyond compliance and quality, the human element is paramount. A well-designed pharmaceutical facility interior considers the comfort, safety, and productivity of its occupants. This focus on human-centric design improves employee morale, reduces errors, and ultimately enhances overall operational efficiency.
- Ergonomic Workstations: Supporting Health and Efficiency:
- Design workstations and laboratory benches with adjustable heights, proper lighting, and readily accessible controls to minimize physical strain and fatigue for personnel.
- Ensure adequate legroom, comfortable seating, and appropriate reach distances for equipment and supplies to promote good posture and reduce the risk of musculoskeletal injuries.
- Integrate noise reduction strategies in high-traffic or equipment-intensive areas to create a more comfortable and less distracting working environment.
- Optimized Workflow and Circulation: Reducing Fatigue, Improving Flow:
- Plan interior layouts that facilitate logical and efficient movement of personnel, materials, and waste, minimizing unnecessary travel distances and bottlenecks.
- Clearly delineate pathways for personnel (e.g., sterile vs. non-sterile zones) and materials to prevent cross-contamination and improve safety.
- Provide sufficient break rooms, changing facilities, and quiet spaces that are easily accessible, promoting employee well-being and mental rejuvenation.
- Lighting and Biophilia: Creating a Positive Environment:
- Utilize a combination of natural daylight and high-quality artificial lighting to create bright, glare-free workspaces that reduce eye strain and improve visual acuity.
- Incorporate elements of biophilic design, such as views of nature, indoor plants (where appropriate and compliant), or natural materials, to enhance employee connection to the natural environment and reduce stress.
- Design for flexibility in lighting controls, allowing occupants to adjust illumination levels to suit their tasks and preferences.
- Safety and Emergency Preparedness: Protecting Lives:
- Ensure clear signage for exits, emergency equipment (fire extinguishers, AEDs), and hazardous areas.
- Design for clear access to safety showers, eyewash stations, and first aid kits.
- Integrate robust communication systems for emergency alerts and evacuation procedures.
- Provide comfortable and safe changing areas for personal protective equipment (PPE) to encourage proper usage and hygiene.
6. Future-Proofing and Adaptability: Designing for Tomorrow’s Innovations
The pharmaceutical industry is constantly evolving, with new therapies, technologies, and manufacturing methods emerging regularly. A truly successful facility interior must be designed with an eye toward the future, ensuring it can adapt to changing demands without requiring costly and disruptive overhauls.
- Modular Design and Flexibility: Ready for Change:
- Incorporate modular components and flexible layouts that can be easily reconfigured or expanded to accommodate new equipment, processes, or increased production volumes.
- Design utility corridors and accessible service areas that allow for easy upgrades, maintenance, and routing of new pipes, conduits, and ventilation systems without disrupting cleanroom operations.
- Consider “ballroom” concepts in cleanrooms where possible, providing open, adaptable spaces that can be fitted with movable walls or temporary partitions to create different classifications as needed.
- Scalability and Expansion Readiness: Growth Without Disruption:
- Plan for potential future expansion during the initial design phase, ensuring that structural elements, utility capacities, and site layout can support additional manufacturing suites or laboratory spaces.
- Design for segregated areas that allow for independent expansion or modification without impacting ongoing operations in other parts of the facility.
- Future-proof IT and automation infrastructure to support emerging technologies like AI, advanced robotics, and enhanced data analytics platforms.
- Embracing Smart Facility Technologies: The Intelligent Pharmaceutical Hub:
- Integrate intelligent building systems (IBS) that allow for centralized monitoring and control of environmental parameters, security, energy consumption, and equipment status.
- Explore the use of robotics and automation for material handling, aseptic filling, and laboratory processes, requiring appropriate floor loading, clear pathways, and robust power infrastructure.
- Design for seamless integration of advanced manufacturing technologies, such as continuous manufacturing platforms, which require specific spatial arrangements and utility connections.
Finding the Right New York Pharmaceutical Facility Interiors Company
Choosing the right partner for your pharmaceutical facility interior project in New York is perhaps the most critical decision you will make. The intricacies of regulatory compliance, the demands of quality control, the imperative for sustainability, and the opportunities of digital delivery necessitate a highly specialized firm. You need a partner with a proven track record, deep understanding of the unique New York regulatory environment, and unparalleled expertise in innovative design and construction tailored for the pharmaceutical sector. Skydome Designs stands out as a leader in this specialized field, having delivered an impressive 859+ pharmaceutical facility interiors assignments across New York and globally over 24+ years. Our commitment to excellence is reflected in our on‑time delivery rate of >98%, underpinned by rigorous multi‑disciplinary reviews, and comprehensive post‑occupancy support that ensures sustained outcomes for our clients.
What to Look for in New York Construction Experts:
- Extensive Experience and Proven Track Record: Look for a company with a significant portfolio of completed pharmaceutical projects in New York and globally. Experience demonstrates a deep understanding of sector-specific challenges and successful project delivery.
- Specialized Expertise: Ensure the company possesses an in-house team of highly specialized architects, engineers, healthcare planners, and project managers who are intimately familiar with pharmaceutical cGMP requirements, cleanroom design, HVAC validation, and process integration.
- Regulatory Acumen: The partner must demonstrate a thorough understanding of FDA, NYSDOH, local building codes, and environmental regulations, with a history of successful regulatory approvals and audits.
- Commitment to Innovation and Sustainability: Look for firms that actively integrate cutting-edge technologies like BIM, VR/AR, and IoT, and prioritize sustainable design practices to achieve both environmental stewardship and operational efficiency.
- End-to-End Service Delivery: A partner offering complete strategy, design, construction, and handover services can streamline the project, ensure seamless coordination, and provide a single point of accountability.
- Transparent Processes and Communication: Demand transparent cost structures, clear milestone-based reporting, and proactive communication to keep you informed at every stage of the project.
- Strong Client References and Post-Occupancy Support: Positive client testimonials and a commitment to ongoing support after project completion are indicators of a reliable and client-focused partner.
End-to-End Delivery in New York: The Skydome Designs Advantage
Choosing a partner that provides end-to-end delivery for pharmaceutical facility interiors is a strategic decision that simplifies complex projects and ensures cohesive, high-quality outcomes. Skydome Designs offers a comprehensive suite of services that covers every phase of your pharmaceutical facility interior project in New York — from initial strategy and conceptual design, through detailed engineering and construction management, all the way to final handover and ongoing post-occupancy support. Our award‑winning team is dedicated to delivering excellence, characterized by transparent costs, rigorous milestone‑based reporting, and an unwavering commitment to your success in New York.
This holistic approach means you benefit from integrated solutions, reduced risks, and faster project timelines. With Skydome Designs, you gain a partner capable of translating your vision into a fully compliant, high-quality, and future-ready pharmaceutical facility, ensuring seamless coordination and superior results from concept to completion.
Ready to embark on your next pharmaceutical facility interior project with a trusted expert? Leverage Skydome Designs‘ proven record of over 859+ successful assignments globally and in New York, with more than 98% on‑time delivery. Contact us today for a consultation to experience award‑winning end‑to‑end delivery.
FAQ: Pharmaceutical Facility Interiors in New York
What are the key regulatory requirements for pharmaceutical facilities in New York?
Key regulatory requirements for pharmaceutical facilities in New York are multi-layered. They primarily include stringent compliance with FDA current Good Manufacturing Practices (cGMP) guidelines (21 CFR Parts 210 and 211), which govern everything from facility design and environmental controls to personnel and material flow. Additionally, facilities must adhere to specific New York State Department of Health (NYSDOH) regulations for pharmaceutical manufacturing and distribution, local building codes, fire safety regulations, and federal and state environmental regulations from the EPA and NYSDEC regarding waste disposal, emissions, and water usage. Strict safety standards set by OSHA and PESH are also mandatory to protect personnel.
How can BIM improve pharmaceutical facility design and construction in New York?
Building Information Modeling (BIM) significantly improves pharmaceutical facility design and construction by enabling better visualization through 3D models, which facilitates early clash detection among architectural, structural, and MEP systems, thereby reducing costly errors and reworks on-site. BIM enhances collaboration among all project stakeholders by providing a centralized, real-time data model, leading to improved communication and coordination. It also supports accurate cost estimation, scheduling, and lifecycle facility management, optimizing performance and reducing operational expenses over the facility’s lifespan. For New York projects, BIM allows for precise planning against complex local regulations and site-specific challenges.
What are the benefits of sustainable design for pharmaceutical facilities in New York?
Sustainable design offers numerous benefits for pharmaceutical facilities in New York. Environmentally, it reduces the facility’s carbon footprint through energy and water conservation, responsible waste management, and the use of eco-friendly materials, aligning with New York’s green initiatives. Economically, it leads to significant long-term operational cost savings due to reduced energy consumption from high-efficiency HVAC and lighting systems, lower water bills from conservation technologies, and minimized waste disposal costs. Furthermore, sustainable design enhances the facility’s corporate image, improves employee well-being through better indoor air quality and natural light, and can offer incentives or tax credits in New York for green building practices.
How do I choose the right pharmaceutical facility interiors company in New York?
Choosing the right pharmaceutical facility interiors company in New York requires careful consideration. Look for a company with extensive, proven experience in pharmaceutical facility design and construction, specifically within the New York regulatory environment. They should have a deep understanding of FDA cGMP, cleanroom classifications, and critical utility systems. Evaluate their in-house expertise (architects, engineers, healthcare planners), relevant certifications, and a strong portfolio of successful projects. Critically, seek a partner offering end-to-end delivery (strategy, design, construction, handover), a transparent approach to costs and reporting, and positive client references, like Skydome Designs, who have delivered 859+ pharmaceutical facility assignments globally and in New York with over 98% on-time delivery.
Why is future-proofing important for pharmaceutical facilities in New York?
Future-proofing is critical for pharmaceutical facilities in New York due to the rapid advancements in medical science, manufacturing technologies, and regulatory landscapes. Designing a facility with adaptability and scalability in mind ensures it can accommodate new processes, equipment, and production demands without requiring extensive, costly, and disruptive renovations. This includes incorporating modular design, flexible utility infrastructure, and robust IT systems that can integrate emerging technologies like AI and robotics. A future-proofed facility remains competitive, compliant, and operationally efficient, securing its long-term value and enabling rapid response to market changes or new therapy development, which is particularly vital in New York’s innovative biotech ecosystem.
Conclusion: Partnering for Excellence in Pharmaceutical Facility Interiors in New York
Navigating the inherent complexities of pharmaceutical facility interiors in New York demands more than just construction; it requires a deep, comprehensive understanding of regulatory requirements, an unwavering commitment to quality control measures, the integration of forward-thinking sustainable design principles, and the strategic adoption of digital delivery methodologies. As we look towards 2025 and beyond, the success of your pharmaceutical venture in New York will be inextricably linked to the foresight and precision embedded within your facility’s interior design and execution.
By meticulously utilizing these comprehensive checklists and, most importantly, by partnering with seasoned and experienced professionals like Skydome Designs, you can confidently ensure your pharmaceutical facility not only meets the highest standards of compliance, quality, and sustainability but also stands as a benchmark for innovation and operational excellence. Our track record of delivering 859+ pharmaceutical facility interiors assignments across New York and globally over 24+ years, coupled with an outstanding on‑time delivery rate of >98% and robust post‑occupancy support, demonstrates our profound capability to underpin truly exceptional outcomes for our clients.
Ready to collaborate with an award‑winning team and create a cutting-edge pharmaceutical facility in New York that is compliant, efficient, and future-ready? Choose Skydome Designs for end‑to‑end delivery for your pharmaceutical facility interiors — encompassing strategy, design, construction, and seamless handover. Our transparent costs and milestone‑based reporting empower you with complete oversight and confidence.
Contact Skydome Designs today for a personalized consultation. Take the first step towards realizing your vision with a partner committed to your success in the dynamic New York pharmaceutical landscape.
Call us directly at +91 7299072144 or email us at info@skydomedesigns.com
Skydome Designs specializes in a broad range of high-stakes interior design and construction projects, demonstrating a versatile expertise that directly informs our pharmaceutical facility work:
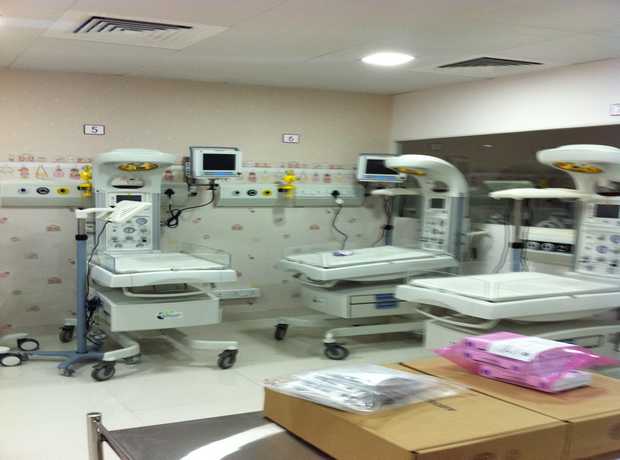
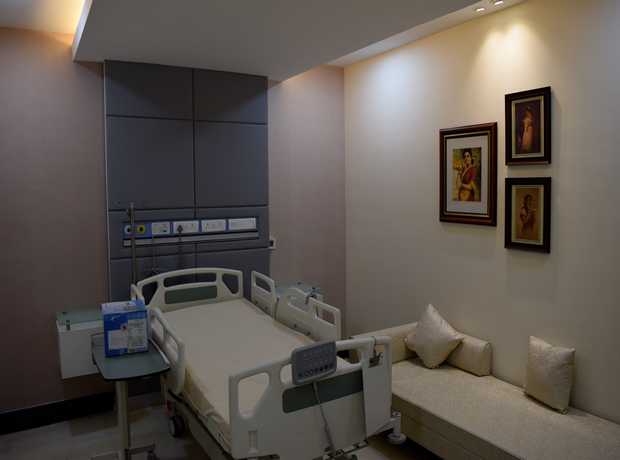
- Hospital Interior Design: Patient rooms, ICUs, OTs, labs, consultation areas, and comprehensive facility planning for optimized care environments.
- Residential Projects: Apartments, luxury condos, senior housing, and community-focused interiors, crafted for comfort and functionality.
- Retail & Commercial Design: Shopping malls, mixed-use developments, corporate offices, and entertainment centers, designed for engaging and efficient user experiences.
- Interior Solutions: Comprehensive space planning, innovative furniture layouts, advanced lighting design, and full turnkey interior execution services, ensuring every detail is perfectly managed.